Play all audios:
ABSTRACT AIM: To investigate the effects of the novel N6-substituted adenosine derivative
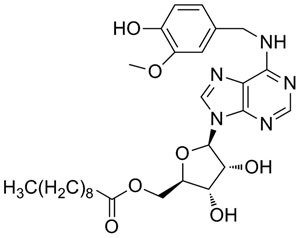
{(2_R_,3_S_,4_R_,5_R_)-3,4-dihydroxy-5-[6-[(4-hydroxy-3-methoxybenzyl)amino]-9_H_-purin-9-yl]tetrahydrofuran-2-yl} methyl decanoate (WS0701) on stress-induced excessive fear, anxiety, and
cognitive deficits in a mouse model of posttraumatic stress disorder (PTSD). METHODS: Male mice underwent a conditioned foot shock and single prolonged stress procedure to induce PTSD.
Contextual/cued fear, elevated plus-maze, open field and novel object recognition tests were conduced to assess PTSD-like behaviors. From d 1, the mice were orally administered WS0701 (7.5,
15, or 30 mg·kg-1·d-1) or paroxetine (10 mg·kg-1·d-1) for two weeks. Apoptosis of hippocampal neurons was detected using flow cytometry and TUNEL staining, and expression of Bcl-2 and Bax in
the hippocampus was measured with Western boltting and qPCR assays. RESULTS: WS0701 administration significantly alleviated fear, anxious behaviors and memory deficits in the mouse model of
PTSD. Furthermore, WS0701 administration significantly reduced the stress-induced apoptosis of hippocampal neurons, and increased the Bcl-2/Bax ratio in the hippocampus. The positive
control drug paroxetine exerted similar effects on PTSD-like behaviors and hippocampal neuron apoptosis in the mouse model of PTSD, which were comparable to those caused by the high dose of
WS0701. CONCLUSION: WS0701 effectively mitigates stress-induced PTSD-like behaviors in mice, partly via inhibition of neuronal apoptosis in the hippocampus. SIMILAR CONTENT BEING VIEWED BY
OTHERS BETA-HYDROXYBUTYRATE, AN ENDOGENOUS NLRP3 INFLAMMASOME INHIBITOR, ATTENUATES ANXIETY-RELATED BEHAVIOR IN A RODENT POST-TRAUMATIC STRESS DISORDER MODEL Article Open access 10 December
2020 BREXPIPRAZOLE BLOCKS POST-TRAUMATIC STRESS DISORDER-LIKE MEMORY WHILE PROMOTING NORMAL FEAR MEMORY Article 19 August 2020 MDMA TREATMENT PAIRED WITH A TRAUMA-CUE PROMOTES ADAPTIVE
STRESS RESPONSES IN A TRANSLATIONAL MODEL OF PTSD IN RATS Article Open access 03 May 2022 INTRODUCTION Individuals exposed to life-threatening trauma are at risk for developing
post-traumatic disorder (PTSD). People who develop PTSD respond to a traumatic experience with intense fear, helplessness or horror and may endure chronic psychological distress due to
repeatedly reliving their trauma through intrusive flashback memories1. This re-experiencing of the trauma can foster the development of debilitating symptoms, such as persistent anxiety,
exaggerated startle responses and cognitive impairments2. These symptoms correlate with the dysregulation of the hypothalamus-pituitary-adrenal (HPA) axis, the dysregulation of several
neurotransmitter systems3 and functional or structural changes in several brain regions4. The hippocampus is one of the most important regions for the development of PTSD and is involved in
explicit memory processes and the encoding of context during fear conditioning. The hippocampus also appears to interact with the amygdala during the encoding of emotional memories, a
process that is highly relevant to the study of trauma and PTSD. In animals, reduced hippocampal volumes and memory impairments can result from extreme stressors, high levels of
stress-related hormones, and abnormal hippocampal function5. The currently used treatments for PTSD include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine
reuptake inhibitors (SNRIs), sedatives, adrenergic blockers, monoamine oxidase inhibitors (MAOIs), atypical anti-psychotics, and anticonvulsants6. Because they have fewer side effects and
are better tolerated, SSRIs are used as the first-line treatment for PTSD. Paroxetine and sertraline are the only Food and Drug Administration (FDA)-approved pharmacotherapies for PTSD, but
these medications are not ideal. Thus, the development of new drugs for the treatment of PTSD is needed. A growing body of evidence suggests that the adenosine system can have multiple
effects such as anticonvulsant, anxiolytic, and sleep-inducing effects, and the adenosine system can inhibit the discharges of amygdala and locus coeruleus and have neuroprotective effects
in some conditions. Adenosine-related drugs have been used for the treatment of anxiety disorder, panic disorder, schizophrenia, epilepsy, and drug addiction7. AMG-1 is an N6 substituted
adenosine derivative extracted from _Armillaria mellea_, which is a traditional Chinese medicine. Previous research has shown that AMG-1 has neuroprotective8 and anticonvulsant effects9 that
are related to reductions in cellular energy metabolism10 and glutamate release11 and inhibition of increases in intracellular calcium12.
{(2_R_,3_S_,4_R_,5_R_)-3,4-dihydroxy-5-[6-[(4-hydroxy-3-methoxybenzyl)amino]-9_H_-purin-9-yl]tetrahydrofuran-2-yl}methyl decanoate (WS0701), the structure of which has been confirmed by
1H-NMR and ESI-MS13 (Figure 1), is a modified form of AMG-1 that was created to increase the sedative effect of AMG-1. PTSD is a severe anxiety disorder, but currently there is little
evidence about the effects of adenosine or its derivatives in the regulation of PTSD in animal models. Therefore, we were interested in whether the adenosine derivative WS0701 would have
effects on the behavioral changes induced in the PTSD mouse model. Furthermore, we explored the protective effects of WS0701 against aversive stress-induced neuronal apoptosis in the
hippocampus. MATERIALS AND METHODS ANIMALS Male KunMing mice were obtained from the CoLab Biotechnology Ltd Laboratory Animal Center (Beijing, China, certificate No SCXK2009-2007). The mice
were clean animals and initially weighed 12±2 g. The mice were housed 6 per cage in a controlled environment with a 12 h light-dark cycle (lights on 7:00–19:00) at 23±2 °C and 45%±10%
humidity. Food and water were available _ad libitum_. Animals received 5 d of habituation to the housing conditions before the stress handling. During the 5-d habituation period, the mice
were handled once daily. Handling consisted of holding the animal with gloved hands for 1 min. All animal experiments were performed during the light phase from 9:00 to 17:00. All animal
treatments were in strict accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The experiments were performed under the approval of
Experimental Animal Administration Committee of Peking Union Medical College. DRUGS AND ADMINISTRATION The drugs used were paroxetine (Seroxat® tablet; GlaxoSmithKline KK, Tianjin, China)
and WS0701 (purity over 98% as determined by HPLC and NMR; provided by the Institute of Materia Medica at Peking Union Medical College, Beijing, China). Both paroxetine and WS0701 were
dissolved in 20% (_w/v_) hydroxypropyl-β-cyclodextrin (HP-β-CD, AR, Sinopharm Chemical Reagent Co, Ltd, Beijing, China) and then diluted to the proper concentration. Animals were randomized
and placed into one of the following 6 groups: the Control, CF+SPS (conditioned foot shock and single prolonged stress), paroxetine (10 mg/kg), or one of three WS0701 (7.5, 15, and 30 mg/kg)
groups. The drugs or vehicle were administered intragastrically once daily 30 min before the experiment from d 1 to d 14. Behavioral assessments were conducted on the corresponding days and
video-recorded for off-line scoring (shown in Figure 2). All behavioral tests were conducted in a sound-proof testing room with constant illumination to which the animals were habituated
for 2 h in their home cages before the initiation of the experiments. All behavioral scoring was performed by two blinded, trained observers. STUDY PROCEDURES EXPERIMENT 1 We conducted
different behavioral tests to study the effects of chronic WS0701 treatment on fear responses, anxiety levels and memory deficits in the stressed mice. The retention of contextual fear was
assessed by observing freezing behavior after re-exposure to trauma-associated cues. Anxiety levels were tested using the elevated plus maze (EPM) and open field (OF) test, and memory
impairment was assessed using novel object recognition (NOR); _n_=12 animals per group (Figure 2). EXPERIMENT 2 To investigate the effects of WS0701 on apoptosis in the hippocampus, brains
were collected under anesthesia on d 14 after the drug treatment for flow cytometry (FCM, _n_=5), terminal deoxynucleotidyl transferase mediated dUTP nick end labeling (TUNEL, _n_=3),
Western blotting (_n_=7) or qPCR (_n_=3) (Figure 2). CONDITIONED FOOT SHOCK AND SINGLE PROLONGED STRESS PROCEDURE For training, we used a Plexiglass chamber (15 cm×15 cm×40 cm) with a
stainless steel grid floor. Mice were placed into the shock chamber; after a 60-s adaptation period, a bright light was turned on in the chamber. After 10 s, the mice received inescapable
electric foot shocks (intensity: 1 mA, duration: 5 s) delivered through the grid floor by an isolated shock generator (YLS-9A, Yiyan Co, Ltd, Ji-nan, China). Control animals underwent the
same procedure but were not subjected to electric foot shocks. This procedure was repeated for 5 consecutive days. A single prolonged stress (SPS) procedure was carried out on the 6th d.
Mice were immobilized for 2 h in a Plexiglas mouse holder device, followed immediately by a 20-min forced swimming test in 24±1 °C water. The forced swim was carried out in a plastic tub (20
cm diameter, 30 cm height) that was two-thirds full of water. Following a 15-min recuperation period, the mice were exposed to ether until they lost consciousness, and they were then
returned to their home cages14,15. CONDITIONED FEAR TEST To study contextual fear, the mice were re-exposed to the shock chamber for 5 min and received a further shock application without
light presentation. To test for cued fear, the mice were placed into the neutral test chamber. After 30 s, a light was presented for 5 min, and the mice were placed back in their home cages
after the tests. “Freezing” behavior was defined as sustained immobility (excluding respiration movements). Fear was quantified as the time (in seconds) that each mouse spent “freezing”14.
ELEVATED PLUS MAZE TEST The EPM apparatus consists of two open and two enclosed arms (30 cm×5 cm) that face each other and are attached to a center platform (5 cm×5 cm) that is elevated 50
cm off the floor. Each mouse was placed on the center platform and was allowed to explore the maze for 5 min. The time spent in and the visits to the open and closed arms during the
observation period were recorded, and the percentage of open arm (OA) entries and the time spent in the OAs were calculated16. OPEN FIELD TEST Each mouse was placed in the center of a cubic
chamber (40 cm×40 cm×35 cm) with a black floor. The floor was divided into sixteen squares (4×4). The testing room was dimly illuminated with indirect white lighting. Each animal was placed
in the chamber for 5 min at a fixed starting point. The distance traveled in the central area and the time spent in the central area (defined as the mouse being in the central squares) were
recorded using a behavioral analysis system (Dig-Behav, Jiliang Co, Shanghai, China)17. NOVEL OBJECT RECOGNITION TEST This experiment consisted of three sessions: the habituation phase (5
min), the training phase (10 min), and the test phase (5 min). In the habituation phase, the mouse was placed into a wooden chamber (50 cm×50 cm×40 cm) for 5 min without an object. Then,
during the training phase, two identical objects were placed near the corners of the chamber (10 cm from each adjacent wall). Each mouse was placed in the center of the floor and allowed to
explore both objects for 10 min. Exploration was defined as the mouse positioning its nose toward the object at a distance of less than 2 cm. The time spent sitting or resting against the
object was not recorded. The test phase occurred four hours after the training phase. In the test phase, the mouse was placed into the box, which contained one familiar object and one novel
object, for 5 min. The exploration time for each object for each animal was collected manually by an experimenter blind to the treatment. The recognition rate (%) was defined as the novel
object exploration time/total exploration time×10018. ANALYSIS OF APOPTOSIS BY FLOW CYTOMETRY The mice were anesthetized with 10% (_w/v_) chloral hydrate and sacrificed by decapitation, and
the hippocampus was dissected. Each sample was placed into cold PBS and made into a single-cell suspension, and the cell concentration was adjusted to 1×104/mL. Next, 1 mL suspension was
taken out and centrifuged at 800 rounds per minute at 4 °C for 5 min, and the supernatant was discarded. Then, 1 mL cold PBS was added and shaken gently to resuspend the cells.
Centrifugation was repeated three times to obtain purified pellets. Cell pellets were resuspended in 500 μL binding buffer and then incubated with 5 μL Annexin V-FITC and 5 μL propidium
iodide while being shielded from light at room temperature for 15 min. Finally, apoptotic cells were analyzed with a FACSCalibur® flow cytometer (BD Biosciences, Burlington, MA, USA).
Approximately 1×104 cells from each sample were analyzed19. IMMUNOHISTOCHEMICAL STAINING After anesthesia with 10% (_w/v_) chloral hydrate, the mice were perfused with 4% paraformaldehyde
(PFA)/PBS (0.01 mol/L), pH 7.2, and the brains were removed. Samples were dehydrated in alcohol and embedded in paraffin. Coronal sections of approximately 2 μm were prepared for
morphological studies. Dewaxed sections were washed three times (5 min each) in 0.01 mol/L PBS and permeabilized in proteinase K for 10 min. Endogenous peroxidase was deactivated by 0.3%
hydrogen peroxide. The sections were washed three more times and were then incubated with TdT at 37 °C for 1 h, followed by incubation with antibody at 37 °C for 1 h. After hematoxylin
post-staining, the sections were stained with 3,3′-diaminobenzidine, mounted, and photographed under a light microscope. The Image-Pro Plus software (Media Cybernetics, Silver Spring, MA,
USA) was used for image analysis. The TUNEL-positive cell rate was calculated as the number of TUNEL-positive cells/total cells×100. Three animals in each group were analyzed, and the mean
TUNEL-positive cell rate was determined20. WESTERN BLOTTING ANALYSIS Seven animals were analyzed in each group. Protein lysates were obtained by homogenization with lysis buffer. Protein
content was determined using the BCA method. Fifty-microgram protein samples were separated on SDS-polyacrylamide gels and transferred onto PVDF membranes. Rabbit Bcl-2 antibody (1:1000) and
rabbit Bax antibodies (1:1000) were used as the primary antibodies (Cell Signaling Technology, Inc, USA). HRP-conjugated anti-rabbit antibodies for Bcl-2 and Bax were used as secondary
antibodies (1:2000, Santa Cruz Biotechnology, Inc, USA). Band detection was performed using an enhanced chemiluminescence detection system (ECL, Molecular Imager ChemiDoc XRS+ System,
Bio-Rad, Hercules, CA, USA). To confirm equal protein amounts, the same blots were incubated with antibodies specific for β-actin (Santa Cruz Biotechnology; 1:2000). The band volumes and
densities were measured using the Quantity One 1D analysis software (Bio-Rad, Hercules, CA, USA). QUANTIFICATION OF HIPPOCAMPAL MRNA EXPRESSION LEVELS Total RNA was extracted using TRIzol®
reagent (Invitrogen Inc, CA, USA), following the manufacturer's instructions. RNA was quantified with optical density measurements at 260 and 280 nm. Reverse transcription was then
performed on 1 μg of total RNA using the SuperScript First-strand Synthesis SuperMix (Invitrogen Inc, CA, USA). The reaction was incubated at 42 °C for 10 min and 95 °C for 2 min. Reverse
transcription products were stored at -80 °C. qPCR was performed on the cDNA samples. The GAPDH gene was used to normalize the expression. qPCR was performed in a volume of 25 μL reaction
mixture that contained 2 μL cDNA, 12.5 μL 2.5×TransStart® Green qPCR SuperMix, 0.5 μL 10 μmol/L forward primer, 0.5 μL 10 μmol/L reverse primer and DNase-free water using a qPCR Kit
(TransGen Inc, CA, USA). The samples were heated to 95 °C for 30 s. The cycles were 95 °C for 5 s, 55 °C for 15 s and 72 °C for 10 s. Relative gene expression was determined using the CFX
Manager® software (Bio-Rad, Inc, Hercules, CA, USA) according to the 2−ΔΔCtmethod21. Additionally, derived melting curve analyses were performed to assess the specificity of the
amplification reaction. The following primers were used: Bcl-2 forward: 5′-ACCGTCGTGACTTGGCAGAG-3′; Bcl-2 reverse: 5′-GGTGTGCAGATGCCGGTTCA-3′; Bax forward: 5′-CGGCGAATTGGAGATGAACTG-3′; Bax
reverse: 5′-GCAAAGTAGAAGAGGGCAACC-3′; GAPDH forward: 5′-TAAAGACCTCTATGCCAACACAGT-3′; and GAPDH reverse: 5′-CACGATGGAGGGGCCGGACTCATC-3′. STATISTICAL ANALYSIS All statistical analyses were
performed using SPSS (Statistical Product and Service Solutions) 20.0 (Chicago, IL, USA). The data are presented as the mean±SD. Statistical analysis included one way analyses of variance
(ANOVAs) followed by Newmann-Keuls tests, when appropriate. For all tests, _P_<0.05 was considered significant. RESULTS CHRONIC WS0701 TREATMENT REDUCED FREEZING BEHAVIOR As shown in
Figure 3, the contextual fear and cued fear of the CF+SPS group [_F_(5,66)=10.288, _P_<0.01; _F_(5,66)=8.220, _P_<0.01] were significantly increased compared with the control group,
and WS0701 administration decreased contextual fear and cued fear compared to the CF+SPS group (15 mg/kg: _P_<0.05; 30 mg/kg: _P_<0.05). CHRONIC WS0701 TREATMENT DECREASED THE
ANIMALS' ANXIETY ELEVATED PLUS MAZE TEST As shown in Figure 4A and 4B, the OA entries and OA times of the CF+SPS group in the EPM were decreased compared to the control group [_F_(5,66)
entries=42.996, _P_<0.001; _F_(5,66) time=20.931, _P_<0.001]. At doses of 7.5, 15, and 30 mg/kg, WS0701 significantly increased the number of OA entries and the OA time compared to
the CF+SPS group (_P_<0.05). OPEN FIELD TEST The open field test results showed that the time spent in the center area and the central traveling distances of the CF+SPS group were
significantly decreased compared to the control group [_F_(5,66) time=10.908, _P_<0.001, _F_(5,66) distance=21.153, _P_<0.001]. The administration of WS0701 (15 and 30 mg/kg;
_P_<0.05) increased the time spent in the central area and the traveling distance in the central area compared to the CF+SPS group (Figure 4C, 4D). CHRONIC WS0701 TREATMENT REDUCED MEMORY
IMPAIRMENTS The mice in the control group spent more time exploring the novel object than the familiar object during the test session [_F_(5,66)=6.582, _P_<0.001]. In contrast, the
stressed mice spent significantly less time exploring the novel object (_P_<0.05), which indicated the presence of memory deficits in this group. Mice treated with WS0701 (15 mg/kg,
_P_<0.05) spent more time exploring the novel object than the mice in the CF+SPS group (Figure 5). CHRONIC WS0701 TREATMENT REDUCED HIPPOCAMPAL APOPTOSIS FLOW CYTOMETRY ASSAY As shown in
Figure 6, we performed Annexin V-FITC/PI double-labeled flow cytometry to assess early apoptosis in the hippocampus. The percentage of early apoptotic cells was increased in the stressed
mice [_F_(5,24)=28.830, _P_<0.01]. Compared with the CF+SPS group, paroxetine (10 mg/kg, _P_<0.05) and WS0701 (15 and 30 mg/kg, _P_<0.05) treatment significantly decreased apoptotic
cell percentages. TUNEL STAINING TUNEL-positive cells were rarely detected in the hippocampal dentate gyri of the control group. In contrast, stress significantly promoted neuronal
apoptosis compared to the control group [_F_(5,12)=17.220, _P_<0.01]. WS0701 (15 and 30 mg/kg, _P_<0.05) and paroxetine (10 mg/kg, _P_<0.05) treatment significantly reduced the
numbers of apoptotic neurons in the dentate gyrus (Figure 7). CHRONIC WS0701 TREATMENT INCREASED THE BCL-2/BAX RATIO IN THE HIPPOCAMPUS Compared to the control group, both Western blotting
and qPCR results showed that the stress procedure increased the expression of Bax [_F_Protein(5,36)=10.724, _F_Gene(5,12)=17.99, _P_<0.05] and decreased the Bcl-2/Bax ratio in the
hippocampus. The administration of paroxetine (10 mg/kg) or WS0701 (15 and 30 mg/kg) reduced the expression of Bax (_P_<0.05) and increased the Bcl-2/Bax ratio, which may be related to
the anti-apoptotic effects of these drugs (Figure 8). DISCUSSION The goal of the present study was to evaluate the long-term effects of the novel adenosine derivative WS0701 on fear, anxiety
and memory impairment in a mouse model of PTSD. Animal models play an important role in the evaluation of novel pharmacotherapeutic agents for PTSD. Previous studies have reported on the
fear conditioning model22 and the single prolonged stress model23. These models mimic different symptoms of PTSD patients and have been widely used in PTSD research. Recent studies have
reported that the combined application of the fear conditioning and SPS procedure can reproduce both the behavioral and endocrine changes of PTSD and that paroxetine has protective effects
in this model; thus, we used paroxetine treatment as a positive control14. With this mouse model of PTSD, we demonstrated that the animals submitted to the stressors not only exhibited
specific avoidance of the context and cues associated with the aversive stress but also avoided entering the center squares and the open arms compared with the control mice. At doses of 15
and 30 mg/kg, WS0701 significantly decreased freezing behavior. In the elevated plus maze and open field tests, WS0701 increased the times spent in the open arms and the central area at all
doses. Moreover, the stressed mice exhibited decreased novel object recognition abilities in a familiar environment, which indicates spatial memory deficits. At a dose of 15 mg/kg, WS0701
prevented the cognitive deficits induced by stress. Though the 30 mg/kg WS0701 group also exhibited some improvement relative to the control groups, this difference did not reach
significance, which may, in part, be due to the stronger sedative effects of WS0701 at this dose. These results showed that WS0701 alleviated the fear responses of stressed animals in the
context associated with the traumatic event and reduced anxiety levels and memory impairments induced by the aversive procedure. PTSD often involves biochemical and molecular changes in the
hippocampus. The results of the flow cytometry and TUNEL experiment indicated that apoptosis in the hippocampus significantly increased in the stressed mice, while the stressed mice treated
with WS0701 (15 and 30 mg/kg) exhibited decreased levels of apoptosis. To reveal the possible mechanisms of action of WS0701, we used Western blotting and qPCR to examine the expression
level of Bax and Bcl-2, which are important mediators of the mitochondria-mediated apoptosis pathway. The Bcl-2/Bax ratio is critical in determining whether apoptosis occurs. The
down-regulation of the Bcl-2/Bax ratio may contribute to the reduced hippocampal volumes and impaired hippocampal function that are associated with PTSD. In this study, both the protein and
gene expression levels of Bcl-2 and Bax were measured. Bcl-2/Bax ratios were significantly reduced in the hippocampi of the stressed mice, while paroxetine and WS0701 (15 and 30 mg/kg)
significantly increased the Bcl-2/Bax ratios in the hippocampi of stressed mice. PTSD is a severe anxiety disorder. Anxiolytics that are used clinically include benzodiazepines, SSRIs,
opiate narcotics and non-benzodiazepine sedatives. SSRIs are currently the most widely used drugs for the treatment of PTSD. Paroxetine is approved by the FDA for the treatment of PTSD, and
it has been reported that paroxetine can reduce PTSD symptoms in animal models24. Paroxetine possibly acts on monoamines in a nonspecific manner, increases neurotrophic factors and thus
exerts neuroprotective effects in the hippocampus. Benzodiazepines are the classic anxiolytics and act on the GABA benzodiazepine receptor to produce sedative, hypnotic, anxiolytic and
anticonvulsant effects. Although these properties suggest a useful role for benzodiazepines in PTSD, benzodiazepines have not been shown to treat PTSD effectively in clinical and animal
studies25. Recent studies have revealed that the opiate analgesic morphine is effective in preventing PTSD in people experiencing physical injuries from traumatic events26. Morphine is an
opioid receptor agonist, and the mechanism by which opioids can inhibit the development of PTSD may be due to pain relief and the blockade of fear memory consolidation; however, the use of
opioids is limited by addiction and abuse problems27. Adenosine has been reported to have anxiolytic, sedative, hypnotic and anticonvulsant effects. It has also been revealed that the
anxiolytic and neuroprotective effects of adenosine are related to the activation of the adenosine A2A receptor28,29. WS0701 was derived from AMG-1, which is structurally similar to
adenosine. In our study, we found that WS0701 produced anxiolytic and neuroprotective effects that are similar to those of adenosine. Therefore, we infer that the mode of action of WS0701
may be related to the activation of the adenosine A2A receptor, and this hypothesis will be studied in our future experiments. CONCLUSION In this study, the repeated administration of WS0701
exhibited a protective effect in a mouse model of PTSD. WS0701 administration reduced the anxiety, fear behaviors and memory deficits induced by aversive stressors, and its mechanism of
action may be related to the inhibition of neuronal apoptosis in the hippocampus. We propose that WS0701 may have potential as a treatment for the prevention of PTSD and possibly other
anxiety disorders after traumatic events. Further experiments are necessary to clarify the relationship between WS0701 and the regulation of emotionality during stressful conditions as well
as the exact molecular mechanisms underlying the protective effects of WS0701. AUTHOR CONTRIBUTION Song WU synthesized and provided WS0701; Guan-hua DU directed the research and revised the
manuscript; Zhong-lin HUANG performed the research and wrote the paper; Rui LIU, Xiao-yu BAI, Gang ZHAO, and Jun-ke SONG revised the manuscript. REFERENCES * American Psychiatric
Association. Diagnostic and statistical manual of mental disorders fourth edition text revision (DSM-IV-TR). Washington DC: American Psychiatric Association; 2000. * Nemeroff CB, Bremner JD,
Foa EB, Mayberg HS, North CS, Stein MB . Posttraumatic stress disorder: a state-of-the-science review. _J Psychiatr Res_ 2006; 40: 1–21. Article Google Scholar * Quirk GJ, Mueller D .
Neural mechanisms of extinction learning and retrieval. _Neuropsychopharmacology_ 2008; 33: 56–72. Article Google Scholar * Nutt DJ . The psychobiology of posttraumatic stress disorder. _J
Clin Psychiatry_ 2000; 61: 24–9. CAS PubMed Google Scholar * Shin LM, Rauch SL, Pitman RK . Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. _Ann N Y Acad Sci_ 2006;
1071: 67–79. Article Google Scholar * Steckler T, Risbrough V . Pharmacological treatment of PTSD - established and new approaches. _Neuropharmacology_ 2012; 62: 617–27. Article CAS
Google Scholar * Lopes LV, Sebastiao AM, Ribeiro JA . Adenosine and related drugs in brain diseases: present and future in clinical trials. _Curr Top Med Chem_ 2011; 11: 1087–101. Article
CAS Google Scholar * Watanabe N, Obuchi T, Tamai M, Araki H, Omura S, Yang JS, _et al_. A novel N6-substituted adenosine isolated from mi huan jun (_Armillaria mellea_) as a
cerebral-protecting compound. _Planta Med_ 1990; 56: 48–52. Article CAS Google Scholar * Chen B, Yang B, Yang LX, Lu ZH . Effects of AMG-1 on the PTZ-kindling seizures in mice. _Neural
Injury and Functional Reconstruction_ 2009; 4: 173–5. Google Scholar * Liu L, Feng YP, Hu D, Li HS, Zhang JT . Effects of AMG-1 on energy metabolism and neuronal damage of ischemic brain in
mice and rats. _Yao Xue Xue Bao_ 1991; 26: 881–5. CAS PubMed Google Scholar * Liu L, Feng YP . Effect of AMG-1 and adenosine on glutamate release from synaptosomes in rats. _Yao Xue Xue
Bao_ 1993; 28: 881–5. CAS PubMed Google Scholar * Li WP, Feng YP, Zhang JT . Effect of AMG-1 on free intrasynaptosome calcium level and on the isolated artery contraction induced by
norepinephrine. _Yao Xue Xue Bao_ 1994; 29: 721–4. CAS PubMed Google Scholar * Wu S, Hao LH, Du GH . Inventors; institute of materia medica, Chinese Academy of Medical Sciences &
Peking Union Medical College, assignee. A new kind of adenosine derivatives and its preparation method. CN patent 200910081639.6. 2009 Apr 07. * Wang H, Zuo D, He B, Qiao F, Zhao M, Wu Y .
Conditioned fear stress combined with single-prolonged stress: a new PTSD mouse model. _Neurosci Res_ 2012; 73: 142–52. Article Google Scholar * Khan S, Liberzon I . Topiramate attenuates
exaggerated acoustic startle in an animal model of PTSD. _Psychopharmacology (Berl)_ 2004; 172: 225–9. Article CAS Google Scholar * Monsef-Esfahani HR, Amini M, Goodarzi N, Saiedmohammadi
F, Hajiaghaee R, Faramarzi MA, _et al_. Coumarin compounds of _Biebersteinia multifida_ roots show potential anxiolytic effects in mice. _Daru_ 2013; 21: 51. Article CAS Google Scholar *
Campos AR, Barros AI, Albuquerque FA, M Leal LK, Rao VS . Acute effects of guarana (_Paullinia cupana_ Mart) on mouse behaviour in forced swimming and open field tests. _Phytother Res_
2005; 19: 441–3. Article CAS Google Scholar * Zhang R, Xue G, Wang S, Zhang L, Shi C, Xie X . Novel object recognition as a facile behavior test for evaluating drug effects in AbetaPP/PS1
Alzheimer's disease mouse model. _J Alzheimers Dis_ 2012; 31: 801–12. Article CAS Google Scholar * Bilsland JG, Haldon C, Goddard J, Oliver K, Murray F, Wheeldon A, _et al_. A rapid
method for the quantification of mouse hippocampal neurogenesis _in vivo_ by flow cytometry. Validation with conventional and enhanced immunohistochemical methods. _J Neurosci Methods_
2006; 157: 54–63. Article Google Scholar * Kyrylkova K, Kyryachenko S, Leid M, Kioussi C . Detection of apoptosis by TUNEL assay. _Methods Mol Biol_ 2012; 887: 41–7. Article CAS Google
Scholar * Ferreira ID, Rosario VE, Cravo PV . Real-time quantitative PCR with SYBR Green I detection for estimating copy numbers of nine drug resistance candidate genes in _Plasmodium
falciparum_. _Malar J_ 2006; 5: 1. Article Google Scholar * Siegmund A, Wotjak CT . A mouse model of posttraumatic stress disorder that distinguishes between conditioned and sensitised
fear. _J Psychiatr Res_ 2007; 41: 848–60. Article Google Scholar * Yamamoto S, Morinobu S, Takei S, Fuchikami M, Matsuki A, Yamawaki S, _et al_. Single prolonged stress: toward an animal
model of posttraumatic stress disorder. _Depress Anxiety_ 2009; 26: 1110–7. Article Google Scholar * Takahashi T, Morinobu S, Iwamoto Y, Yamawaki S . Effect of paroxetine on enhanced
contextual fear induced by single prolonged stress in rats. _Psychopharmacology_ 2006; 189: 165–73. Article CAS Google Scholar * Ipser JC, Stein DJ . Evidence-based pharmacotherapy of
post-traumatic stress disorder (PTSD). _Int J Neuropsychopharmacol_ 2012; 15: 825–40. Article CAS Google Scholar * Holbrook TL, Galarneau MR, Dye JL, Quinn K, Dougherty AL . Morphine use
after combat injury in Iraq and post-traumatic stress disorder. _N Engl J Med_ 2010; 362: 110–7. Article CAS Google Scholar * Szczytkowski-Thomson JL, Lebonville CL, Lysle DT . Morphine
prevents the development of stress-enhanced fear learning. _Pharmacol Biochem Behav_ 2013; 103: 672–7. Article CAS Google Scholar * Chen JF, Sonsalla PK, Pedata F, Melani A, Domenici MR,
Popoli P, _et al_. Adenosine A2A receptors and brain injury: broad spectrum of neuroprotection, multifaceted actions and “fine tuning” modulation. _Prog Neurobiol_ 2007; 83: 310–31. Article
CAS Google Scholar * Alsene K, Deckert J, Sand P, de Wit H . Association between A2a receptor gene polymorphisms and caffeine-induced anxiety. _Neuropsychopharmacology_ 2003; 28:
1694–702. Article CAS Google Scholar Download references ACKNOWLEDGEMENTS This study was supported by the International S&T Cooperation Projects (No 2009DFA32010), the National
Scientific and Technological Major Project (2009ZX09102-003 and 2013ZX09J13102-05C), and the Special Foundation for Public Welfare Industry of Health (No 200902008), China. AUTHOR
INFORMATION AUTHORS AND AFFILIATIONS * Beijing Key Laboratory of Drug Targets Identification and Drug Screening, Institute of Materia Medica, Chinese Academy of Medical Sciences and Peking
Union Medical College, Beijing, 100050, China Zhong-lin Huang, Rui Liu, Xiao-yu Bai, Gang Zhao, Jun-ke Song & Guan-hua Du * Department of New Drug Research and Development, State Key
Laboratory of Bioactive Substance and Function of Natural Medicines, Institute of Materia Medica, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100050, China
Song Wu & Guan-hua Du Authors * Zhong-lin Huang View author publications You can also search for this author inPubMed Google Scholar * Rui Liu View author publications You can also
search for this author inPubMed Google Scholar * Xiao-yu Bai View author publications You can also search for this author inPubMed Google Scholar * Gang Zhao View author publications You can
also search for this author inPubMed Google Scholar * Jun-ke Song View author publications You can also search for this author inPubMed Google Scholar * Song Wu View author publications You
can also search for this author inPubMed Google Scholar * Guan-hua Du View author publications You can also search for this author inPubMed Google Scholar CORRESPONDING AUTHOR
Correspondence to Guan-hua Du. POWERPOINT SLIDES POWERPOINT SLIDE FOR FIG. 1 POWERPOINT SLIDE FOR FIG. 2 POWERPOINT SLIDE FOR FIG. 3 POWERPOINT SLIDE FOR FIG. 4 POWERPOINT SLIDE FOR FIG. 5
POWERPOINT SLIDE FOR FIG. 6 POWERPOINT SLIDE FOR FIG. 7 POWERPOINT SLIDE FOR FIG. 8 RIGHTS AND PERMISSIONS Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Huang, Zl., Liu, R.,
Bai, Xy. _et al._ Protective effects of the novel adenosine derivative WS0701 in a mouse model of posttraumatic stress disorder. _Acta Pharmacol Sin_ 35, 24–32 (2014).
https://doi.org/10.1038/aps.2013.143 Download citation * Received: 11 April 2013 * Accepted: 28 August 2013 * Published: 16 December 2013 * Issue Date: January 2014 * DOI:
https://doi.org/10.1038/aps.2013.143 SHARE THIS ARTICLE Anyone you share the following link with will be able to read this content: Get shareable link Sorry, a shareable link is not
currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt content-sharing initiative KEYWORDS * posttraumatic stress disorder * WS0701 * adenosine *
AMG-1 * paroxetine * fear * anxiety * memory deficits * hippocampus * apoptosis * Bcl-2 * Bax