Play all audios:
Sevoflurane, in particular multiple exposures, has been reported to cause the abnormal neurological development including attention-deficit/hyperactivity disorder (ADHD). This study is to
investigate ADHD-like impulsivity in adult mice after repeated sevoflurane exposures at the neonatal stage. Six-day-old pups were exposed to 60% oxygen in the presence or absence of 3%
sevoflurane for 2 h and the treatment was administrated once daily for three consecutive days. To assess the impulsivity, the cliff avoidance reaction (CAR) was carried out at the 8th week.
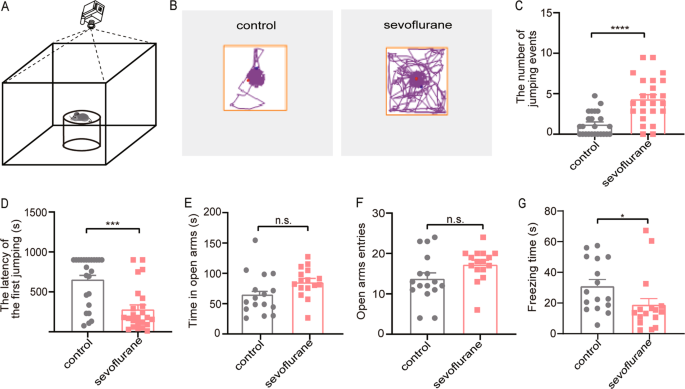
Our results showed that repeated sevoflurane treatment increased the number of jumps and shortened the jumping latency in the CAR test. The cortices were harvested for immunostaining to
detect c-Fos and calmodulin-dependent protein kinase IIα (CaMKIIα) expression in the medial prefrontal cortex (mPFC). We found that mPFC neurons, especially excitatory neurons, were highly
activated and related to impulsive behavior. The activation viruses (AAV-CaMKIIα-hM3Dq) were injected to evaluate the effects of specific activation of mPFC excitatory neurons on impulsive
behavior in the presence of clozapine-N-oxide (CNO). Likewise, the inhibitory viruses (AAV-CaMKIIα-hM4Di) were injected in the sevoflurane group to explore whether the mPFC excitatory
neuronal inhibition reduced the impulsivity. Our results revealed that chemogenetic activation of mPFC excitatory neurons induced impulsive behavior whereas inhibition of mPFC excitatory
neurons partially rescued the deficit. These results indicate that repeated sevoflurane exposures at the critical time induce impulsive behavior accompanied with overactivation of mPFC
excitatory neurons in adult stages. This work may further extend to understand the ADHD-like impulsive behavior of the anesthetic neurotoxicity.
Attention-deficit/hyperactivity disorder (ADHD) is a chronic neurodevelopmental disorder manifesting primarily as attention deficit, hyperactivity, and impulsivity1. ADHD affects 5–8% of
school-age children, in which 50–80% cases persist till adulthood2. Adult ADHD has complex manifestations. Currently, the core symptom used in adult ADHD diagnosis is impulsivity, defined as
poorly conceived control deficit3,4. Impulsivity increases the risk of pathological gambling, substance abuse, and relationship failure. As the prevalence rate of adult ADHD is soaring over
the world, therefore, neurobiological and psychosocial factors related to the etiology and symptom control of ADHD have been studied in depth—exemplified by genes, neurotransmitters, neural
pathways, drugs, and education.
Patients with ADHD show remarkable variation in complicating factors and neuropsychological weaknesses5. Previous studies suggested that the impaired development of dopaminergic neurons is
related to ADHD development6. In addition to disorders in neurotransmitter system, other studies indicated that ADHD is caused by abnormal structural and functional synapses in mPFC6,7,
which is responsible for behavioral inhibition and executive functions. Recently, clinical studies reported that repeated exposures to inhalational anesthetics in children is associated with
the development of ADHD6,8,9,10. This finding was further supported by an animal study, which revealed that ketamine administration caused ADHD-like hyperactive behavior11. However, whether
anesthetics have a long-term effect on ADHD development, particularly impulsive behavior, remains uncertain. Given the potential issue of public mental health and social stability,
identifying the links between general anesthesia and impulse control disorder is now of paramount importance. Sevoflurane, the most commonly used pediatric anesthetic, has been shown to act
as a neurotoxin affecting synaptogenesis and neuronal morphology in animal studies12,13. Therefore, we hypothesize that exposure of the immature brain to sevoflurane may induce ADHD-like
behavior conditions in later adulthood.
In this study, we exposed neonatal mice to sevoflurane and found it induced ADHD-like impulsive behavior in the cliff avoidance reaction (CAR) test in adult age. We revealed that sevoflurane
permanently affected the mPFC excitatory neurons, which were involved in impulsive behavior. We also demonstrated that chemogenetic inhibition of the mPFC excitatory neurons rescued
sevoflurane-induced impulsive behavior. These results suggest that sevoflurane-induced impulsive behavior is another type of long-term outcome to anesthetic neurotoxicity. Furthermore,
excitatory neuronal activation in the mPFC may serve as a pathological mechanism for anesthetics-induced ADHD in adults.
All procedures were performed following the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals and approved by the Animal Advisory Committee at Zhejiang
University. C57BL/6J mice were group-housed in a temperature-controlled room at the Animal Facility of Zhejiang University under a 12 h/12 h light/dark cycle. Food and water were available
ad libitum. Male and female mice were allowed to meet for one night and then the females were housed individually. Time mated birth was monitored and pups were collected for further
experiments.
Animal groups were determined in a pilot study and randomly assigned to the sevoflurane group and control group before the experiment. At postnatal day 6 (P6), pups were placed in a
translucent plastic chamber (20 × 10 × 10 cm3) with a layer of soda-lime and wet cotton on the bottom floor. The temperature of the chamber floor was maintained at 32 °C using a heating pad
under the chamber. The chamber was connected to a sevoflurane vaporizer (Vapor 2000; Medical Systems, Inc., Germany) and continuously flushed with 60% oxygen balanced nitrogen (FIO2 = 0.6)
at 2 L/min for 2 h with (the sevoflurane group) or without (the control group) 3% sevoflurane. The treatment was administrated once per day for 3 consecutive days. Mice were allowed to grow
up to 8 weeks. Female mice were excluded to avoid the possible confounding effects of the estrous cycle and only males were used for further experiments.
One cohort of mice (n = 24) was evaluated for the CAR test. CAR is a behavioral paradigm used to investigate the ADHD-like impulsive behavior in mature rodents14,15,16. It depends on the
natural tendency of animals to avoid falling from a height, which is more than twice the length of the animal17. On account of a control deficit and risk-taking choice, the impulsive mice
were more active in exploring the cliff, which led to a jump from the high platform at a higher frequency. According to a previous study18, a round platform (an inverted glass beaker with 11
cm diameter and 15 cm height) placed in the center of the open field apparatus (45 × 45 × 45 cm3) was used for the CAR test. The behavioral experiments were conducted in a quiet room
illuminated by dim light between 9 and 12 am. Before the tests, the mice (n = 24) were handled in a friendly manner once/day for 3 consecutive days to decrease the stress-related neuronal
activation. On the test day, mice were first allowed to habituate in the test room for more than 30 min. Each mouse was then gently placed on the platform and the duration that it remained
on the platform (from the initial placement until falling onto the floor) was recorded. During the whole 15 min testing period, the mice were repeatedly placed on the platform once they fell
off, and the cumulative cliff-falling events were documented. The latency is defined as the duration when mice stayed on the platform within the 15 min testing period. The apparatus was
cleaned with 75% ethanol between tests to prevent olfactory influence.
Another cohort of mice (n = 16) were evaluated for the EPM test. The EPM consists of two open arms and two closed arms (30 × 5 cm2) in an arrangement that the two arms of each type are
opposite to each other. The maze was placed at the height of 65 cm above the ground. A video camera was fixed above the maze to record the movements of mice. Each mouse was placed in the
center area facing the open arms and allowed to explore freely for 5 min. Time spent in the open arms, the number of entries into the open arms and freezing time were calculated using the
video-based ANY-maze system (Stoelting, USA). The apparatus was cleaned with 75% ethanol between tests.
90 min after completing the CAR test, 6 mice were sacrificed under terminated anesthesia the CAR test according to a random number table, and then perfused with 4% paraformaldehyde (PFA).
Their brains were harvested for immunohistochemistry analysis. Coronal brain sections (40-μm thickness) were cut by a freezing microtome (CM30503, Leica Biosystems, Germany). After 2-h
blocking in 5% normal donkey serum (NDS; 017-000-121, Jackson ImmunoResearch Laboratories, Inc. USA), 1% bovine serum albumin (BSA; A2153, Sigma-Aldrich, USA), and 0.3% Triton X-100
(Sigma-Aldrich, USA), the sections were incubated with the primary antibodies (mouse anti–c-Fos [1:1,000; Abcam, ab208942] and/or rabbit anti–CaMKIIα [1:1000; Abcam, ab52476]) overnight at 4
°C. Brain sections were then incubated with secondary antibodies (Alexa Fluor 488 anti-mouse IgG [1:1,000; A-21202, Thermo Fisher Scientific] or Alexa Fluor 546 anti-rabbit IgG [1:1,000;
A-10040, Thermo Fisher Scientific]). The processed brain sections were then mounted onto glass slides with 60% glycerol in PBS. Images were captured using a Nikon A1 laser-scanning confocal
microscope (Nikon, Japan). The number of immune-positive cells was counted and analyzed by ImageJ (NIH, USA). DAPI (4’,6-2- phenylindole; 1:3,000; C1002; Beyotime) was used simultaneously to
identify nuclei.
Under sodium pentobarbital anesthesia (50 mg/kg, intraperitoneal injection (i.p.)), other cohorts were mounted in a stereotaxic apparatus (RWD Life Science, Shenzhen, China). After adjusting
the position of the skull horizontally under a stereomicroscope (RWD Life Science, Shenzhen, China)19, a small craniotomy was made with a thin drill over the medial prefrontal cortex
(typical coordinate: 1.9 mm anterior to Bregma; 0.34 mm lateral to the midline). Adeno-associated virus (AAV) carrying fusion genes for hM3D (AAV- CaMKIIα-hM3D(Gq)-mCherry), hM4D (AAV-
CaMKIIα-hM4D(Gi)-mCherry), or mcherry (AAV-CaMKIIα -mcherry) were injected using a 10 µL micro-syringe (#701, Hamilton, USA) at a rate of 60 nL/min by micro-syringe pump (kdScientific, USA).
Over a 10-min period, 80 nL viruses were injected at a depth of 2.4 mm from the Bregma. Mice with viral injections in the incorrect area or expressions beyond the targeted area (mPFC) were
excluded from the study. The hM3Dq was injected unilaterally for activation (n = 7) and hM4Di (n = 8) was bilaterally injected for inhibition. The control adenovirus (mCherry only) was also
injected unilaterally or bilaterally for corresponding control experiments. The viruses hM3Dq (AAV2/9, 3.6 × 1012 genomic copies per ml), hM4Di (AAV2/9, 3.6 × 1012 genomic copies per mL), or
mcherry (AAV2/9, 3.54 × 1012genomic copies per mL) were made by HanBio (Shanghai, China).
The mice injected with the above viruses were received a saline i.p. to test the baseline of the CAR deficit. After that, mice were given CNO (1 mg/kg i.p.; clozapine-N-oxide; HY17366,
MedchemExpress, USA) for virus-infected neuron manipulation.
After a 3–4-week viral incubation, coronal sections including the mPFC were cut at a thickness of 300 μm using a vibratome (VT 1200S, Leica, Germany) in ice-cold cutting solution (234 mM
Sucrose, 5 mM KCl, 1.25 mM NaH2PO4, 5 mM MgSO4, 26 mM NaHCO3, 25 mM dextrose, 1 mM CaCl2, oxygenated with 95% O2/ 5% CO2) as described previously19. Next, slices were incubated in the
aerated (95% O2/5% CO2) artificial cerebrospinal fluid (ACSF: 124 mM NaCl, 2 mM KCl, 1.25 mM KH2PO4, 2 mM MgSO4, 2 mM CaCl2, 26 mM NaHCO3, and 10 mM D-(C) glucose, pH 7.4.) to recover for 30
min at 32 °C, and subsequently incubated for 1 h at room temperature. Brain slices were then transferred to a recording chamber. To test function of expressed hM3Dq or hM4Di protein,
whole-cell patch-clamp recordings were applied with glass pipettes backfilled with an artificial intracellular fluid (120 mM K-gluconate, 15 mM KCl, 10 mM HEPES, 4 mM Mg-ATP, 0.3 mM
Tris-GTP, 0.5 mM EGTA, adjusted to pH 7.3 with KOH, 285–290 mOsm). Pipettes were connected to the headstage of a Heka EPC 10 amplifier (Heka Elektronik, USA). Cells expressing viral-encoded
fluorescent markers (mCherry) were visualized under microscope with infrared differential interference contrast optics (BX51WI, Olympus, Japan). Current-clamp recording was used to measure
evoked action potentials in both CNO activation and inhibition experiment. After applying currents in steps of 20 pA (from 0 to 360 pA) neurons were recovered for 5 min. The brain slices
were then perfused with ACSF containing 5 µM CNO. The same current-clamp procedure was performed 10 min after CNO perfusion.
The sample size was estimated by software PASS.11 (NCSS, USA) according to the preliminary experimental results. Experienced researchers conducted independently the different experiments and
blinded data collectors analyzed these results. All data are shown as mean ± SEM unless otherwise specified. Before analysis, all data underwent the Kolmogorov–Smirnov normality test.
Comparisons between two groups were made with unpaired or paired Student’s t-test if data are normally distributed; otherwise, Mann–Whitney U test or Wilcoxon signed-rank tests were used.
Variance was found to be similar between the groups as tested using the Levene test of homogeneity of variances. Two-way ANOVA followed by Bonferroni’s post-hoc test was used for multiple
comparisons, as appropriate. p-value less than 0.05 was considered to be statistically significant, defined as *p