Play all audios:
ABSTRACT Macrophages present a spectrum of phenotypes that mediate both the pathogenesis and resolution of atherosclerotic lesions. Inflammatory macrophage phenotypes are pro-atherogenic,
but the stimulatory factors that promote these phenotypes remain incompletely defined. Here we demonstrate that microbial small RNAs (msRNA) are enriched on low-density lipoprotein (LDL) and
drive pro-inflammatory macrophage polarization and cytokine secretion via activation of the RNA sensor toll-like receptor 8 (TLR8). Removal of msRNA cargo during LDL re-constitution yields
particles that readily promote sterol loading but fail to stimulate inflammatory activation. Competitive antagonism of TLR8 with non-targeting locked nucleic acids was found to prevent
native LDL-induced macrophage polarization in vitro, and re-organize lesion macrophage phenotypes in vivo, as determined by single-cell RNA sequencing. Critically, this was associated with
reduced disease burden in distinct mouse models of atherosclerosis. These results identify LDL-msRNA as instigators of atherosclerosis-associated inflammation and support alternative
functions of LDL beyond cholesterol transport. Access through your institution Buy or subscribe This is a preview of subscription content, access via your institution ACCESS OPTIONS Access
through your institution Access Nature and 54 other Nature Portfolio journals Get Nature+, our best-value online-access subscription $29.99 / 30 days cancel any time Learn more Subscribe to
this journal Receive 12 print issues and online access $209.00 per year only $17.42 per issue Learn more Buy this article * Purchase on SpringerLink * Instant access to full article PDF Buy
now Prices may be subject to local taxes which are calculated during checkout ADDITIONAL ACCESS OPTIONS: * Log in * Learn about institutional subscriptions * Read our FAQs * Contact customer
support SIMILAR CONTENT BEING VIEWED BY OTHERS TREM2 PROMOTES FOAMY MACROPHAGE LIPID UPTAKE AND SURVIVAL IN ATHEROSCLEROSIS Article Open access 30 October 2023 E3 UBIQUITIN LIGASE RNF128
PROMOTES LYS63-LINKED POLYUBIQUITINATION ON SRB1 IN MACROPHAGES AND AGGRAVATES ATHEROSCLEROSIS Article Open access 04 March 2025 ABERRANT MITOCHONDRIAL DNA SYNTHESIS IN MACROPHAGES
EXACERBATES INFLAMMATION AND ATHEROSCLEROSIS Article Open access 26 August 2024 CODE AVAILABILITY Informatics tools used for sequencing analysis in this manuscript are available for public
use via GitHub (https://github.com/shengqh). Additional support is available through the corresponding authors. REFERENCES * Danesh, J., Collins, R., Appleby, P. & Peto, R. Association
of fibrinogen, C-reactive protein, albumin, or leukocyte count with coronary heart disease: meta-analyses of prospective studies. _JAMA_ 279, 1477–1482 (1998). Article CAS Google Scholar
* Ridker, P. M., Rifai, N., Rose, L., Buring, J. E. & Cook, N. R. Comparison of C-reactive protein and low-density lipoprotein cholesterol levels in the prediction of first
cardiovascular events. _N. Engl. J. Med._ 347, 1557–1565 (2002). Article CAS Google Scholar * Sabatine, M. S. et al. Evolocumab and clinical outcomes in patients with cardiovascular
disease. _N. Engl. J. Med._ 376, 1713–1722 (2017). Article CAS Google Scholar * Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. _N. Engl. J.
Med._ 377, 1119–1131 (2017). Article CAS Google Scholar * Moore, K. J. & Tabas, I. Macrophages in the pathogenesis of atherosclerosis. _Cell_ 145, 341–355 (2011). Article CAS Google
Scholar * Moore, K. J. & Freeman, M. W. Scavenger receptors in atherosclerosis: beyond lipid uptake. _Arterioscler. Thromb. Vasc. Biol._ 26, 1702–1711 (2006). Article CAS Google
Scholar * Babaev, V. R. et al. Reduced atherosclerotic lesions in mice deficient for total or macrophage-specific expression of scavenger receptor-A. _Arterioscler. Thromb. Vasc. Biol._ 20,
2593–2599 (2000). Article CAS Google Scholar * Suzuki, H. et al. A role for macrophage scavenger receptors in atherosclerosis and susceptibility to infection. _Nature_ 386, 292–296
(1997). Article CAS Google Scholar * Que, X. et al. Oxidized phospholipids are proinflammatory and proatherogenic in hypercholesterolaemic mice. _Nature_ 558, 301–306 (2018). Article CAS
Google Scholar * Febbraio, M. et al. Targeted disruption of the class B scavenger receptor CD36 protects against atherosclerotic lesion development in mice. _J. Clin. Invest._ 105,
1049–1056 (2000). Article CAS Google Scholar * Stewart, C. R. et al. CD36 ligands promote sterile inflammation through assembly of a toll-like receptor 4 and 6 heterodimer. _Nat.
Immunol._ 11, 155–161 (2010). Article CAS Google Scholar * Miller, Y. I. Toll-like receptors and atherosclerosis: oxidized LDL as an endogenous toll-like receptor ligand. _Future
Cardiol._ 1, 785–792 (2005). Article CAS Google Scholar * Steinbrecher, U. P. & Lougheed, M. Scavenger receptor-independent stimulation of cholesterol esterification in macrophages by
low density lipoprotein extracted from human aortic intima. _Arterioscler. Thromb. J. Vasc. Biol._ 12, 608–625 (1992). Article CAS Google Scholar * Steinberg, D. & Witztum, J. L. Is
the oxidative modification hypothesis relevant to human atherosclerosis? Do the antioxidant trials conducted to date refute the hypothesis? _Circulation_ 105, 2107–2111 (2002). Article
Google Scholar * Kim, Kyeongdae et al. Transcriptome analysis reveals nonfoamy rather than foamy plaque macrophages are proinflammatory in atherosclerotic murine models. _Circ. Res._ 123,
1127–1142 (2018). Article CAS Google Scholar * Fernandez, D. M. et al. Single-cell immune landscape of human atherosclerotic plaques. _Nat. Med._ 25, 1576–1588 (2019). Article CAS
Google Scholar * Zernecke, Alma et al. Meta-analysis of leukocyte diversity in atherosclerotic mouse aortas. _Circ. Res._ 127, 402–426 (2020). Article CAS Google Scholar * Spann, N. J.
et al. Regulated accumulation of desmosterol integrates macrophage lipid metabolism and inflammatory responses. _Cell_ 151, 138–152 (2012). Article CAS Google Scholar * Jaiswal, S. et al.
Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. _N. Engl. J. Med._ 377, 111–121 (2017). Article Google Scholar * Estruch, M. et al. CD14 and TLR4 mediate cytokine
release promoted by electronegative LDL in monocytes. _Atherosclerosis_ 229, 356–362 (2013). Article CAS Google Scholar * Lamarche, B., Lemieux, I. & Després, J. P. The small, dense
LDL phenotype and the risk of coronary heart disease: epidemiology, patho-physiology and therapeutic aspects. _Diabetes Metab._ 25, 199–211 (1999). CAS Google Scholar * Smith, E. B. &
Ashall, C. Low-density lipoprotein concentration in interstitial fluid from human atherosclerotic lesions. Relation to theories of endothelial damage and lipoprotein binding. _Biochim.
Biophys. Acta_ 754, 249–257 (1983). Article CAS Google Scholar * Kruth, H. S., Huang, W., Ishii, I. & Zhang, W.-Y. Macrophage foam cell formation with native low density lipoprotein.
_J. Biol. Chem._ 277, 34573–34580 (2002). Article CAS Google Scholar * Goldstein, J. L., Ho, Y. K., Basu, S. K. & Brown, M. S. Binding site on macrophages that mediates uptake and
degradation of acetylated low density lipoprotein, producing massive cholesterol deposition. _Proc. Natl Acad. Sci. USA._ 76, 333–337 (1979). Article CAS Google Scholar * Anzinger, J. J.
et al. Native low-density lipoprotein uptake by macrophage colony-stimulating factor-differentiated human macrophages is mediated by macropinocytosis and micropinocytosis. _Arterioscler.
Thromb. Vasc. Biol._ 30, 2022–2031 (2010). Article CAS Google Scholar * Jackson, W. D., Weinrich, T. W. & Woollard, K. J. Very-low and low-density lipoproteins induce neutral lipid
accumulation and impair migration in monocyte subsets. _Sci. Rep._ 6, 20038 (2016). Article CAS Google Scholar * Goulopoulou, S., McCarthy, C. G. & Webb, R. C. Toll-like receptors in
the vascular system: sensing the dangers within. _Pharmacol. Rev._ 68, 142–167 (2016). Article Google Scholar * Seimon, T. A. et al. Atherogenic lipids and lipoproteins trigger
CD36-TLR2-dependent apoptosis in macrophages undergoing endoplasmic reticulum stress. _Cell Metab._ 12, 467–482 (2010). Article CAS Google Scholar * Mistry, P. et al. Inhibition of TLR2
signaling by small molecule inhibitors targeting a pocket within the TLR2 TIR domain. _Proc. Natl Acad. Sci. USA._ 112, 5455–5460 (2015). Article CAS Google Scholar * Kadl, A. et al.
Identification of a novel macrophage phenotype that develops in response to atherogenic phospholipids via Nrf2. _Circ. Res._ 107, 737–746 (2010). Article CAS Google Scholar * Vaeth, M. et
al. Ca2+ signaling but not store-operated Ca2+ entry is required for the function of macrophages and dendritic cells. _J. Immunol._ 195, 1202–1217 (2015). Article CAS Google Scholar *
Heil, F. et al. Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. _Science_ 303, 1526–1529 (2004). Article CAS Google Scholar * Tanji, H. et al.
Toll-like receptor 8 senses degradation products of single-stranded RNA. _Nat. Struct. Mol. Biol._ 22, 109–115 (2015). Article CAS Google Scholar * Sarvestani, S. T. et al.
Sequence-dependent off-target inhibition of TLR7/8 sensing by synthetic microRNA inhibitors. _Nucleic Acids Res._ 43, 1177–1188 (2015). Article CAS Google Scholar * Hornung, V. et al.
Sequence-specific potent induction of IFN-α by short interfering RNA in plasmacytoid dendritic cells through TLR7. _Nat. Med._ 11, 263–270 (2005). Article CAS Google Scholar * Zhang, S.
et al. Small-molecule inhibition of TLR8 through stabilization of its resting state. _Nat. Chem. Biol._ 14, 58–64 (2018). Article CAS Google Scholar * Jurk, M. et al. Human TLR7 or TLR8
independently confer responsiveness to the antiviral compound R-848. _Nat. Immunol._ 3, 499 (2002). Article CAS Google Scholar * Diebold, S. S., Kaisho, T., Hemmi, H., Akira, S. &
Reis e Sousa, C. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. _Science_ 303, 1529–1531 (2004). Article CAS Google Scholar * Liu, J. et al. A
five-amino-acid motif in the undefined region of the TLR8 ectodomain is required for species-specific ligand recognition. _Mol. Immunol._ 47, 1083–1090 (2010). Article CAS Google Scholar
* Ishii, N., Funami, K., Tatematsu, M., Seya, T. & Matsumoto, M. Endosomal localization of TLR8 confers distinctive proteolytic processing on human myeloid cells. _J. Immunol._ 193,
5118–5128 (2014). Article CAS Google Scholar * Gorden, K. K. B., Qiu, X. X., Binsfeld, C. C. A., Vasilakos, J. P. & Alkan, S. S. Cutting edge: activation of murine TLR8 by a
combination of imidazoquinoline immune response modifiers and polyT oligodeoxynucleotides. _J. Immunol._ 177, 6584–6587 (2006). Article CAS Google Scholar * Allen, R. M. et al.
Bioinformatic analysis of endogenous and exogenous small RNAs on lipoproteins. _J. Extracell. Vesicles_ 7, 1506198 (2018). Article CAS Google Scholar * Krieger, M., Brown, M. S., Faust,
J. R. & Goldstein, J. L. Replacement of endogenous cholesteryl esters of low density lipoprotein with exogenous cholesteryl linoleate. Reconstitution of a biologically active lipoprotein
particle. _J. Biol. Chem._ 253, 4093–4101 (1978). Article CAS Google Scholar * Jonsson, A. L. & Bäckhed, F. Role of gut microbiota in atherosclerosis. _Nat. Rev. Cardiol._ 14, 79–87
(2017). Article CAS Google Scholar * Robbins, M. et al. 2’-O-methyl-modified RNAs act as TLR7 antagonists. _Mol. Ther. J. Am. Soc. Gene Ther._ 15, 1663–1669 (2007). Article CAS Google
Scholar * Sioud, M., Furset, G. & Cekaite, L. Suppression of immunostimulatory siRNA-driven innate immune activation by 2′-modified RNAs. _Biochem. Biophys. Res. Commun._ 361, 122–126
(2007). Article CAS Google Scholar * Mitchell, S. et al. An NFκB activity calculator to delineate signaling crosstalk: type I and II interferons enhance NFκB via distinct mechanisms.
_Front. Immunol_. 10, Article 1425 (2019). * Rayner, K. J. et al. Antagonism of miR-33 in mice promotes reverse cholesterol transport and regression of atherosclerosis. _J. Clin. Invest._
121, 2921–2931 (2011). Article CAS Google Scholar * Rahman, S. M. et al. CCAAT/Enhancer-binding protein β (C/EBPβ) expression regulates dietary-induced inflammation in macrophages and
adipose tissue in mice. _J. Biol. Chem._ 287, 34349–34360 (2012). Article CAS Google Scholar * Zernecke, A. & Weber, C. Chemokines in atherosclerosis: proceedings resumed.
_Arterioscler. Thromb. Vasc. Biol._ 34, 742–750 (2014). Article CAS Google Scholar * Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol
crystals. _Nature_ 464, 1357–1361 (2010). Article CAS Google Scholar * Sugiyama, Y., Sugiyama, F., Yagami, K., Miyaji, S. & Kurosawa, T. Comparison of plasma endotoxin levels in
germ-free, SPF and conventional laboratory animals (mice and rats). _Jikken Dobutsu_ 42, 89–92 (1993). CAS Google Scholar * Wright, S. D. et al. Infectious agents are not necessary for
murine atherogenesis. _J. Exp. Med._ 191, 1437–1442 (2000). Article CAS Google Scholar * Stepankova, R. et al. Absence of microbiota (germ-free conditions) accelerates the atherosclerosis
in ApoE-deficient mice fed standard low cholesterol diet. _J. Atheroscler. Thromb._ 17, 796–804 (2010). Article CAS Google Scholar * Patinote, C. et al. Agonist and antagonist ligands of
toll-like receptors 7 and 8: ingenious tools for therapeutic purposes. _Eur. J. Med. Chem._ 193, 112238 (2020). Article CAS Google Scholar * Janssen, H. L. A. et al. Treatment of HCV
infection by targeting microRNA. _N. Engl. J. Med._ 368, 1685–1694 (2013). Article CAS Google Scholar * He, T. C. et al. A simplified system for generating recombinant adenoviruses.
_Proc. Natl Acad. Sci. USA_ 95, 2509–2514 (1998). Article CAS Google Scholar * Michell, D. L. et al. Isolation of high-density lipoproteins for non-coding small rna quantification. _J.
Vis. Exp._ https://doi.org/10.3791/54488 (2016). * Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. _Can. J. Biochem Physiol._ 37, 911–917 (1959).
Article CAS Google Scholar * Paigen, B., Morrow, A., Holmes, P. A., Mitchell, D. & Williams, R. A. Quantitative assessment of atherosclerotic lesions in mice. _Atherosclerosis_ 68,
231–240 (1987). Article CAS Google Scholar * Linton, M. F., Atkinson, J. B. & Fazio, S. Prevention of atherosclerosis in apolipoprotein E-deficient mice by bone marrow
transplantation. _Science_ 267, 1034–1037 (1995). Article CAS Google Scholar * Makowski, L. et al. Lack of macrophage fatty-acid-binding protein aP2 protects mice deficient in
apolipoprotein E against atherosclerosis. _Nat. Med._ 7, 699–705 (2001). Article CAS Google Scholar * Babaev, V. R. et al. Macrophage EP4 deficiency increases apoptosis and suppresses
early atherosclerosis. _Cell Metab._ 8, 492–501 (2008). Article CAS Google Scholar * Chen, Y., Yu, Q. & Xu, C.-B. A convenient method for quantifying collagen fibers in
atherosclerotic lesions by ImageJ software. _Int J Clin Exp Med._ 10, 14904–14910 (2017). * Park, E. K. et al. Optimized THP-1 differentiation is required for the detection of responses to
weak stimuli. _Inflamm. Res._ 56, 45–50 (2007). Article CAS Google Scholar * Fleetwood, A. J., Lawrence, T., Hamilton, J. A. & Cook, A. D. Granulocyte-macrophage colony-stimulating
factor (CSF) and macrophage csf-dependent macrophage phenotypes display differences in cytokine profiles and transcription factor activities: implications for CSF blockade in inflammation.
_J. Immunol._ 178, 5245–5252 (2007). Article CAS Google Scholar * Na, Y. R., Jung, D., Gu, G. J. & Seok, S. H. GM-CSF grown bone marrow derived cells are composed of phenotypically
different dendritic cells and macrophages. _Mol. Cells_ 39, 734 (2016). CAS Google Scholar * Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. _EMBnet.
J._ 17, 10–12 (2011). Article Google Scholar * Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. _Bioinformatics_ 29, 15–21 (2013). Article CAS Google Scholar * Liao, Y.,
Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. _Bioinformatics_ 30, 923–930 (2014). Article CAS Google
Scholar * Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. _Genome Biol._ 15, 550 (2014). Article Google Scholar *
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. _Genome Biol._ 10, R25 (2009). Article Google
Scholar * Galkina, E. et al. Lymphocyte recruitment into the aortic wall before and during development of atherosclerosis is partially L-selectin dependent. _J. Exp. Med._ 203, 1273–1282
(2006). Article CAS Google Scholar * Liu, Q. et al. scRNABatchQC: multi-samples quality control for single cell RNA-seq data. _Bioinformatics_ 35, 5306–5308 (2019). Article CAS Google
Scholar * Stuart, T. et al. Comprehensive integration of single-cell data. _Cell_ 177, 1888–1902.e21 (2019). Article CAS Google Scholar * Franzén, O., Gan, L.-M. & Björkegren, J. L.
M. PanglaoDB: a web server for exploration of mouse and human single-cell RNA sequencing data. _Database_ 2019, 1–9 (2019). * Audzevich, T. et al. Pre/pro-B cells generate macrophage
populations during homeostasis and inflammation. _Proc. Natl Acad. Sci. USA_ 114, E3954–E3963 (2017). Article CAS Google Scholar * Williams, J. W. et al. Limited proliferation capacity of
aortic intima resident macrophages requires monocyte recruitment for atherosclerotic plaque progression. _Nat. Immunol._ 21, 1194–1204 (2020). Article CAS Google Scholar * Tacke, R. et
al. The transcription factor NR4A1 is essential for the development of a novel macrophage subset in the thymus. _Sci. Rep._ 5, 10055 (2015). Article Google Scholar * Sarrazy, V. et al.
Maintenance of macrophage redox status by ChREBP limits inflammation and apoptosis and protects against advanced atherosclerotic lesion formation. _Cell Rep._ 13, 132–144 (2015). Article
CAS Google Scholar * Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data.
_Bioinformatics_ 26, 139–140 (2010). Article CAS Google Scholar * Greenwald, R. A. _CRC Handbook of Methods for Oxygen Radical Research_ (CRC Press, 1987). Download references
ACKNOWLEDGEMENTS The authors thank W. Reichard, M. Kuzmich, S. Landstreet, A. Ifram, L. Sedgeman, C. Wiese and V. Babaev for technical assistance and helpful discussions. We also thank Q.
Liu of the Vanderbilt Center for Quantitative Sciences for consultation on single-cell sequencing analysis, and A. Jones of VANTAGE at VUMC for expertise in high-throughput sequencing
technologies, the Vanderbilt Flow Cytometry Shared Resource and Translational Pathology Shared Resource. This work is supported by American Heart Association awards 19CDA34660280 (R.M.A.)
and 18IPA34180005 (R.M.A.), W.M. Keck Research Foundation Grant (K.C.V., R.M.A., M.F.L. and Q.S.) and National Institutes of Health grants P01HL116263 (M.F.L.) and R01HL128996 (K.C.V.).
AUTHOR INFORMATION Author notes * Ryan M. Allen Present address: Department of Physiology and Cell Biology, University of Arkansas for Medical Sciences, Little Rock, AR, USA * Kasey C.
Vickers Present address: Department of Medicine, Vanderbilt University Medical Center, Nashville, TN, USA AUTHORS AND AFFILIATIONS * Department of Medicine, Vanderbilt University Medical
Center, Nashville, TN, USA Ryan M. Allen, Danielle L. Michell, Ashley B. Cavnar, Wanying Zhu, Neil Makhijani, Danielle M. Contreras, Chase A. Raby, Mark Castleberry, Youmin Zhang, Amanda C.
Doran, MacRae F. Linton & Kasey C. Vickers * Department of Molecular Physiology and Biophysics, Vanderbilt University, Nashville, TN, USA Elizabeth M. Semler & Kasey C. Vickers *
Department of Biomedical Engineering, Vanderbilt University, Nashville, TN, USA Carlisle DeJulius & Craig Duvall * Department of Biostatistics, Vanderbilt University Medical Center,
Nashville, TN, USA Marisol Ramirez-Solano, Shilin Zhao & Quanhu Sheng Authors * Ryan M. Allen View author publications You can also search for this author inPubMed Google Scholar *
Danielle L. Michell View author publications You can also search for this author inPubMed Google Scholar * Ashley B. Cavnar View author publications You can also search for this author
inPubMed Google Scholar * Wanying Zhu View author publications You can also search for this author inPubMed Google Scholar * Neil Makhijani View author publications You can also search for
this author inPubMed Google Scholar * Danielle M. Contreras View author publications You can also search for this author inPubMed Google Scholar * Chase A. Raby View author publications You
can also search for this author inPubMed Google Scholar * Elizabeth M. Semler View author publications You can also search for this author inPubMed Google Scholar * Carlisle DeJulius View
author publications You can also search for this author inPubMed Google Scholar * Mark Castleberry View author publications You can also search for this author inPubMed Google Scholar *
Youmin Zhang View author publications You can also search for this author inPubMed Google Scholar * Marisol Ramirez-Solano View author publications You can also search for this author
inPubMed Google Scholar * Shilin Zhao View author publications You can also search for this author inPubMed Google Scholar * Craig Duvall View author publications You can also search for
this author inPubMed Google Scholar * Amanda C. Doran View author publications You can also search for this author inPubMed Google Scholar * Quanhu Sheng View author publications You can
also search for this author inPubMed Google Scholar * MacRae F. Linton View author publications You can also search for this author inPubMed Google Scholar * Kasey C. Vickers View author
publications You can also search for this author inPubMed Google Scholar CONTRIBUTIONS R.M.A.: conceptualization, methodology, investigation, formal analysis, visualization and
writing—original draft. D.L.M.: methodology, investigation, formal analysis and writing—reviewing and editing. A.B.C.: investigation and formal analysis. N.M.: investigation and formal
analysis. E.M.S.: formal analysis and investigation. D.M.C.: resources and formal analysis. W.Z.: resources. C. DeJulius: resources. M.C.: resources. Y.Z.: resources. C.A.R.: formal
analysis. M.R.-S.: software and visualization. S.Z. software and visualization. C. Duvall: methodology. A.C.D.: methodology and writing—reviewing and editing. Q.S.: methodology, software,
visualization and writing—reviewing and editing. M.F.L.: methodology, supervision and writing—reviewing and editing. K.C.V.: conceptualization, methodology, supervision, formal analysis,
visualization and writing—reviewing and editing. CORRESPONDING AUTHORS Correspondence to Ryan M. Allen or Kasey C. Vickers. ETHICS DECLARATIONS COMPETING INTERESTS M.F.L. has received
research funding from Amgen, Regeneron, Ionis, Merck, REGENXBIO, Sanofi and Novartis, and has served as a consultant for Esperion, Alexion Pharmaceuticals and REGENXBIO. All other authors
have no competing interests. PEER REVIEW PEER REVIEW INFORMATION _Nature Cell_ Biology thanks Jeffrey Kroon and the other, anonymous, reviewer(s) for their contribution to the peer review of
this work. Peer reviewer reports are available. ADDITIONAL INFORMATION PUBLISHER’S NOTE Springer Nature remains neutral with regard to jurisdictional claims in published maps and
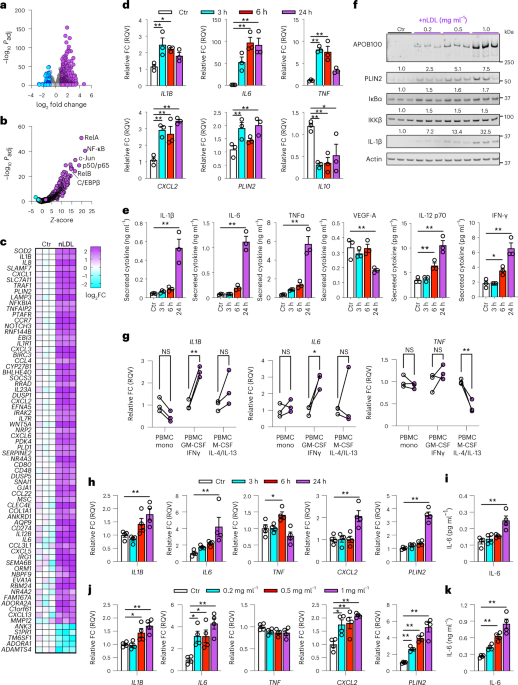
institutional affiliations. EXTENDED DATA EXTENDED DATA FIG. 1 NLDL INDUCES INFLAMMATORY ACTIVATION OF MACROPHAGES. (a) mRNA expression determined by qPCR of THP-1 macrophages treated with
indicated doses of nLDL (matched to Fig. 1f) for 24 h (n = 3 biological replicates). (b) Quantification of immunoblots presented in Fig. 1f (n = 3 biological replicates). (c) Secreted
cytokines in the media of THP-1 macrophages stimulated with 0.5 mg/ml nLDL for 3 h, 6 h, 24 h relative to cells receiving no treatment for 24 h (Ctr) (matched to Fig. 1e; n = 3 biological
replicates) (d) Primary mouse bone-marrow derived macrophages (BMDM) differentiated with GM-CSF were treated with nLDL (0.5 mg/ml) for 24 h and assayed for mRNA expression by qPCR (n = 3
biological replicates), (e) cytokine secretion by ELISA (n = 3 biological replicates), and (f) protein expression in cell lysates by immunoblot (n = 2 biological replicates). Data are mean ±
SEM. (a-c) One-way ANOVA and (d) Two-way ANOVA with Benjamini, Krieger and Yekutieli FDR (Q = 0.05), *q < 0.05, **q < 0.01, ***q < 0.001, ****q < 0.0001. e, Student’s t-test
(unpaired, two-sided), **p < 0.01. Numerical source data, statistics, exact _p_ values and _q_ values are provided. Source data EXTENDED DATA FIG. 2 NLDL AND MACROPHAGE TLR RESPONSES
QUALITY CONTROL. (a) Total protein (top) and neutral lipid (bottom) of ox LDL, bovine serum albumin (BSA), and 10 human nLDL of independent donors resolved by agarose gel electrophoresis.
Image represents two independent experiments. (b) TBARS assay of nLDL samples, or matched LDL samples treated with copper sulfate as indicated (limit of detection = 0.625 μM) (n = 10
independent preparations). (c) Quantification of total protein and lipids of DGUC-VLDL, -LDL, -HDL, or BSA following fractionation with 2x-Superose-6 columns. Lipoprotein data are matched to
a single donor representative of >10 independent experiments. (d) mRNA expression of primary human macrophages (CD14 + ; GM-CSF/IFNγ) pre-treated with C29 (200 μM) for 30 min, then
stimulated with PAM3CSK4 (2 ng/mL) for 4 h (n = 3 biological replicates(BR)). (e) Normalized NF-κB-driven luciferase activity of HEK293T cells over expressing an empty vector, hTLR7 or hTLR8
following treatments with vehicle (Ctr, n = 4 BR), nLDL (0.5 mg/ml, n = 4 BR), ssRNA40 (TLR8 ligand; 2 μg/mL, n = 4 BR), R848 (TLR7 ligand; 10 μM, n = 3 BR), or CL075 (TLR8 ligand; 2.5
μg/mL, n = 3 BR). (f-h) mRNA expression of THP-1 macrophages electroporated with siRNA against _TLR7_,_TLR8_, or no siRNA (Control, n = 4 BR) and then treated with (f) nLDL (0.5 mg/mL, n = 6
BR), (g) R848 (10 μM, n = 6 BR) or (h) ssRNA40 (0.5 μg/mL, n = 6 BR). (i) Relative NF-κB-driven luciferase activity of HEK293T cells over expressing an empty vector (n = 7–8), mTLR7 (n =
7–8) or mTLR8 (n = 6-8) treated with mock transfection, R848 (10 μM), ssRNA40 (2 μg/mL), or nLDL (0.5 mg/ml) for 24 h. (j) mRNA expression in wild-type (WT) and _Tlr7_-/- BMDMs following
treatment with 0.5 mg/ml nLDL, or 1 μg/mL ssRNA40 for 24 h (n = 3 BR). (k) Relative NF-κB-driven luciferase activity of HEK293T cells over expressing human or mTLR8 pre-treated with CU-CPT9a
and exposed to nLDL for 24 h (n = 4 BR). Data are mean ± SEM. (d) Two-way ANOVA; Sidak’s multiple comparisons test, ***p < 0.001, ****p < 0.0001. (e, i-k) Two-way ANOVA; Benjamini,
Krieger and Yekutieli FDR (Q = 0.05), *q < 0.05, **q < 0.01, ***q < 0.001). (f-h) One-way ANOVA; Dunnett’s multiple comparisons test. *p < 0.05, **p < 0.01, ***p < 0.001,
****p < 0.0001. Numerical source data, statistics, exact _p_ values and _q_ values are provided. Source data EXTENDED DATA FIG. 3 THE SMALL RNA ON LDL IS PREDOMINANTLY EXOGENOUS AND
REMOVED BY LDL RE-CONSTITUTION. a) Normalized abundance of taxa identified upon alignment of LDL-sRNA to non-host tRNA database (tRNA-db). RPM, reads per million total reads. b) Normalized
abundance of bacterial phyla (human microbiome database) contributing sRNA to LDL. c) Normalized abundance of fungal sRNA and representative genomes present on LDL. d) Normalized abundance
of algal and protist sRNA and representative genomes present on LDL. RPM, reads per million total reads. Matched nLDL and rLDL samples were fractionated by size-exclusion chromatography
(SEC) using two superose-6 columns in tandem and assessed for e) phospholipid and protein content by colorimetric kit (representative data of three independent experiments), f) fluorescence
(TopFluor Cholesteryl ester), and g) APOB protein by immunoblot (representative image of three independent experiments). h) Relative expression of exogenous sRNA in matched rLDL and nLDL of
a single preparation relative to buffer controls. i) Oil-Red-O staining and fluorescence microscopy (TopFluor Cholesterol ester) (representative images of three biological replicates). Scale
bar = 200 μm. Numerical source data, statistics, exact _p_ values and _q_ values are provided. Source data EXTENDED DATA FIG. 4 MICROBIAL SMALL RNA ON LIPOPROTEINS IS NOT DEPLETED IN
GERM-FREE MICE. a) Plasma from two cohorts of adult mice - specific pathogen free (SPF; n = 6 mice total) and facility-matched germ-free (GF; n = 17 mice total) fed a chow diet were
harvested at the National Gnotobiotic Rodent Resource Center (NGRRC; North Carolina, USA) for lipoprotein sRNA-seq b) Plasma was fractionated by size-exclusion chromatography (SEC) and
cholesterol-rich fractions corresponding with HDL were selected for sRNA-seq. c) Relative percentage of reads aligned to host and non-host databases, as well as reads too short for analysis
or reads that failed to align to either database (unmapped). d) Percentage of sRNA reads aligned to host miRNA, host tRNA and host rRNA transcripts. e) Percentage of reads aligned to the
non-host rRNA database and tRNA database. f) Percentage of reads aligned to genomes of fungi and algae. g) Percentage of reads aligned to bacterial genomes associated with a human microbiome
(HMB) database. h) Reads per million total reads (RPM) mapped to indicated bacterial phyla within the HMB database. i) Percentage of reads aligned to bacterial genomes within an
environmental bacteria (ENV) database. j) Differential abundance (log2) of bacterial sRNA (dots represent individual genomes of the HMB and ENV databases) between GF and SPF mice categorized
by phyla. Gray bar represents a 1.5 fold change. Data are mean ± SEM. (d-g, i) Statistical differences between GF and SPF were assessed by Mann-Whitney U-test, but no evaluations were
statistically significant. j) Differences in abundance of individual genomes within each database were assessed between groups by the Wald Test, but applying False Discovery Rate correction
(α = 0.05) resulted in no differentially abundant genomes. Numerical source data, statistics, exact _p_ values and _q_ values are provided. Source data EXTENDED DATA FIG. 5 LOCKED NUCLEIC
ACID (LNA) BASES MEDIATE ANTAGONISM OF SINGLE-STRANDED RNA LIGANDS OF TLR8. a) HEK293T cells over-expressing human TLR8, UNC93B1 and CD14 were pre-treated with vehicle (DOTAP) or
corresponding DNA/LNA oligonucleotides (2.5 μg/mL) for 30 min and then stimulated with TLR8 nucleoside analogue agonist CL075 or TLR8 ORN agonists ssRNA40 or ORN06 (2 μg/mL) for 24 h (n = 5
biological replicates). Two-way ANOVA with Dunnett’s multiple comparison test (statistical significance relative to untreated within each group; **p < 0.0001). b) THP-1 macrophages were
pre-treated + /- nt-LNA (1 µg/mL) for 45 min and treated with LPS (500 ng/mL), Poly I:C (1 µg/mL), CL075 (2.5 μg/ml), or ssRNA40 at 1 µg/mL (1:1, nt-LNA:ssRNA40) or 0.2 µg/ml (5:1) for 24 h
(n = 3 biological replicates). Relative mRNA expression of _IL1B_, _IL6_ and _TNF_ were then assessed by qPCR. For each treatment, the relative fold change of each treatment in the presence
of nt-LNA was expressed as a percentage of the relative fold change of each treatment without nt-LNA pre-treatment (% inhibition). Two-way ANOVA, Benjamini, Krieger and Yekutieli FDR (Q =
0.05), **q < 0.01. c-d) Primary human CD14 + PBMC differentiated with GM-CSF and IFNγ were pre-treated with 2.5 μg/mL nt-LNA or vehicle (DOTAP) for 30 minutes and then stimulated with or
without ssRNA40 (0.5 μg/mL) for 24 h. c) mRNA expression was quantified by qPCR (n = 4 biological replicates) d) Cytokine (IL-6) secretion was quantified by ELISA (n = 4 biological
replicates). One-way ANOVA with Dunnett’s multiple comparison test. **p < 0.01. ***p < 0.001, ****p < 0.0001 (e) mRNA expression (n = 4 biological replicates), (f) cytokine
secretion (n = 3 biological replicates) and (g) immunoblotting (representative image of three independent experiments) of BMDMs following up to 24 h treatment with IFNγ (100 U/mL) +/- 0.5
mg/ml nLDL in the presence or absence of 2.5 μg/mL nt-LNA. Two-way ANOVA, Benjamini, Krieger and Yekutieli FDR (Q = 0.05), *q < 0.05, **q < 0.01. Data are mean ± SEM. Numerical source
data, statistics, exact _p_ values and _q_ values are provided. Source data EXTENDED DATA FIG. 6 NT-LNA TREATMENT REDUCES ATHEROSCLEROSIS WITHOUT ALTERING LIPID OR LIPOPROTEIN METABOLISM IN
_APOE_-/- MICE. a) Female and male _Apoe_-/- mice fed a western diet were administered saline (Ctr; n = 4 mice per sex), nt-LNA-A (20 mg/kg; n = 4 mice per sex) or nt-LNA-B (20 mg/kg; n = 4
mice per sex) by intraperitoneal injection once weekly for four weeks. Treatments for each were randomized between cohabitating animals separated by sex. At sacrifice, the aortic sinus was
serially sectioned and stained with Oil-Red O to identify atherosclerotic lesions. Scale bar = 500 μm. b) Quantification of lesion area in serial sections and c) sex-normalized, relative
lesion area under the curve (n = 8 mice per treatment). d) Plasma of female _Apoe_-/- mice treated for 4 weeks with saline (Ctr; n = 10) or nt-LNA (n = 10) were fractionated by
size-exclusion chromatography and assessed for total cholesterol (TC) e) Plasma protein levels were assessed by immunoblot of individual cages receiving either Saline/Ctr (n = 5 mice) or
nt-LNA (n = 5 mice) treatments (representative images of two independent assessments). f) Quantification of independent immunoblots by densitometry (n = 10 mice per treatment) normalized to
C3. g) Lesion area (Oil-red O) of matched sections of the aortic root following treatment with saline (Ctr) or nt-LNA for 4 weeks (n = 10 mice per treatment). h) Hepatic mRNA expression
determined by qPCR (n = 10 mice per treatment). Data are mean ± SEM. (c) One-way ANOVA, Sidak’s multiple comparison test, *p < 0.05. **p < 0.01. (f-h) Two-way ANOVA with Benjamini,
Krieger and Yekutieli FDR (Q = 0.05), *q < 0.05, **q < 0.01,***q < 0.001. Numerical source data, statistics, exact _p_ values and _q_ values are provided. Source data EXTENDED DATA
FIG. 7 NT-LNA TREATMENT PROMOTES ATHEROSCLEROTIC REGRESSION IN _LDLR_-/- MICE. (a) Schematic for regression study design. Male(M) and female(F) _Ldlr_-/- mice were fed a chow diet (n = 6
mice) or an atherogenic diet (n = 50 mice) for 14 weeks. After 14 weeks, chow-fed mice and a subset of mice from the atherogenic diet group (baseline; n = 7 M/8 F mice). Remaining diet-fed
mice were then switched to a chow diet to allow lesion regression (Reg.) and were injected once weekly with saline control (Reg. Ctr; n = 9 M/9 F mice) or Reg. nt-LNA (30 mg/kg; n = 9 M/8 F
mice). b) Plasma total cholesterol (TC) or c) triglycerides (TG) following fractionation by SEC; chow (n = 6; 3 M/3 F), baseline (n = 8; 4 M/4 F), Reg. Ctr; (n = 10; 5 M/5 F) and Reg. nt-LNA
(n = 10; 5 M/5 F) d) Immunoblots of plasma proteins in Reg. Ctr (n = 10) or Reg. nt-LNA (n = 9) groups. Representative images of two independent experiments are shown. e) Quantification of
immunoblots by densitometry. f-g) Lesion area of serial sections of the aortic root in baseline (n = 15 mice; 7 M/8 F), Reg. Ctr(n = 18 mice; 9 M/9 F) or nt-LNA (n = 17; 9 M/8 F) groups. h)
Lesion area under the curve (AUC) for both sexes of mice as determined by Oil Red O staining in the aortic root (Baseline: n = 15; Reg. Ctr: n = 18; Reg. nt-LNA; n = 17). One-way ANOVA;
Dunnett’s multiple comparison test, **p < 0.01, ***p < 0.001. i) Lesion AUC for mice of each group separated by sex. Two-way ANOVA; Dunnett’s multiple comparison test, **p < 0.01.
j-k) Masson’s Trichrome staining and quantification of fibrosis in aortic roots of baseline (n = 7 mice; 3 M/4 F) Reg. Ctr (n = 9 mice; 4 M/5 F) or Reg. nt-LNA (n = 10 mice; 5 M/5 F) groups.
l-m) MAC2 (green) immunofluorescence and quantification within aortic roots obtained of at baseline (n = 7 mice; 3 M/4 F), Reg. Ctr (n = 10 mice; 5 M/5 F), or Reg. nt-LNA (n = 10 mice; 5
M/5 F) groups. Two-way ANOVA; Tukey’s multiple comparison test, **p < 0.01, ***p < 0.001. Data are mean ± SEM. Scale bar = 500 μm. Numerical source data, statistics, exact _p_ values
and _q_ values are provided. Source data EXTENDED DATA FIG. 8 GATING STRATEGY OF LEUKOCYTES FROM MOUSE AORTAS FOR SINGLE-CELL RNA SEQUENCING. Sequential gating fluorescent activated cell
sorting for single and live cells, followed by non-red blood cells. Cells were then sorted that were CD45 + but CD3−. EXTENDED DATA FIG. 9 SINGLE-CELL RNA SEQUENCING OF THE ATHEROSCLEROTIC
LESION TO IDENTIFY ANTI-ATHEROSCLEROTIC MECHANISMS OF NT-LNA TREATMENT. a) _Apoe__-/-_ mice fed an atherogenic diet for 4 weeks were injected once weekly with saline control (Ctr; n = 8) or
nt-LNA (30 mg/kg; n = 8). b) UMAP projection of unbiased clusters obtained from atherosclerotic lesions. c) Relative contribution of cells from saline (Ctr) and nt-LNA treated mice to each
cluster of (b). d) Relative expression (color) and % of cells reaching threshold of detection (size) of transcripts pertaining to T cell and NK cell phenotypes (top) or B-cell phenotypes
(bottom) in atherosclerosis for each cluster. Numerical source data, statistics, exact _p_ values and _q_ values are provided. Source data SUPPLEMENTARY INFORMATION REPORTING SUMMARY PEER
REVIEW FILE SUPPLEMENTARY TABLES Supplementary Tables 1–14. SOURCE DATA SOURCE DATA FIG. 1 Numerical source data. SOURCE DATA FIG. 1 Uncropped Western blots. SOURCE DATA FIG. 2 Numerical
source data. SOURCE DATA FIG. 2 Uncropped Western blots. SOURCE DATA FIG. 3 Numerical source data. SOURCE DATA FIG. 4 Numerical source data. SOURCE DATA FIG. 4 Uncropped Western blots.
SOURCE DATA FIG. 5 Numerical source data. SOURCE DATA FIG. 6 Numerical source data. SOURCE DATA FIG. 7 Numerical source data. SOURCE DATA EXTENDED DATA FIG. 1 Numerical source data. SOURCE
DATA EXTENDED DATA FIG. 1 Uncropped Western blots. SOURCE DATA EXTENDED DATA FIG. 2 Numerical source data. SOURCE DATA EXTENDED DATA FIG. 2 Uncropped Western blots. SOURCE DATA EXTENDED DATA
FIG. 3 Numerical source data. SOURCE DATA EXTENDED DATA FIG. 3 Uncropped Western blots. SOURCE DATA EXTENDED DATA FIG. 4 Numerical source data. SOURCE DATA EXTENDED DATA FIG. 5 Numerical
source data. SOURCE DATA EXTENDED DATA FIG. 5 Uncropped Western blots. SOURCE DATA EXTENDED DATA FIG. 6 Numerical source data. SOURCE DATA EXTENDED DATA FIG. 6 Uncropped Western blots.
SOURCE DATA EXTENDED DATA FIG. 7 Numerical source data. SOURCE DATA EXTENDED DATA FIG. 7 Uncropped Western blots. SOURCE DATA EXTENDED DATA FIG. 9 Numerical source data. RIGHTS AND
PERMISSIONS Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s);
author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law. Reprints and permissions ABOUT THIS
ARTICLE CITE THIS ARTICLE Allen, R.M., Michell, D.L., Cavnar, A.B. _et al._ LDL delivery of microbial small RNAs drives atherosclerosis through macrophage TLR8. _Nat Cell Biol_ 24,
1701–1713 (2022). https://doi.org/10.1038/s41556-022-01030-7 Download citation * Received: 22 December 2020 * Accepted: 18 October 2022 * Published: 06 December 2022 * Issue Date: December
2022 * DOI: https://doi.org/10.1038/s41556-022-01030-7 SHARE THIS ARTICLE Anyone you share the following link with will be able to read this content: Get shareable link Sorry, a shareable
link is not currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt content-sharing initiative