Play all audios:
ABSTRACT Standard and direct membrane-feeding assays (SMFA and DMFA) are fundamental assays to evaluate efficacy of transmission-blocking intervention (TBI) candidates against _Plasmodium
falciparum_ and _vivax_. To compare different candidates precisely, it is crucial to understand the error range of measured activity, usually expressed as percent inhibition in either oocyst
intensity (% transmission reducing activity, %TRA), or in prevalence of infected mosquitoes (% transmission blocking activity, %TBA). To this end, mathematical models have been proposed for
_P. falciparum_ SMFA (PfSMFA), but such study for DMFA is limited. In this study, we analyzed _P. vivax_ DMFA (PvDMFA) data from 22,236 mosquitoes tested from 96 independent assays. While
the two assays are quite different, a zero-inflated negative binomial (ZINB) model could reasonably explain the PvDMFA results, as it has for PfSMFA. Our simulation studies based on the ZINB
model revealed it is better to report %TRA values with a proper error range, rather than observed %TBA both in SMFA and DMFA. Furthermore, the simulations help in designing a better assay
and aid in estimating an error range of a %TRA value when the uncertainty is not reported. This study strongly supports future TBI development by providing a rational method to compare
different candidates. SIMILAR CONTENT BEING VIEWED BY OTHERS PREDICTING THE PUBLIC HEALTH IMPACT OF A MALARIA TRANSMISSION-BLOCKING VACCINE Article Open access 08 March 2021 TRANSMISSIBILITY
OF A NEW _PLASMODIUM FALCIPARUM_ 3D7 BANK FOR USE IN MALARIA VOLUNTEER INFECTION STUDIES EVALUATING TRANSMISSION BLOCKING INTERVENTIONS Article Open access 16 April 2025 CONTRASTING VECTOR
COMPETENCE OF THREE MAIN EAST AFRICAN _ANOPHELES_ MALARIA VECTOR MOSQUITOES FOR _PLASMODIUM FALCIPARUM_ Article Open access 17 January 2025 INTRODUCTION According to the World Malaria Report
2019, 228 million clinical cases and 405,000 deaths were estimated due to malaria in 20181. Of the five _Plasmodium_ species which infect humans, _P. falciparum_ is the most abundant and
deadly species, especially for < 5-year old children who live in sub-Saharan Africa regions. _P. vivax_ malaria is the second major contributor to global clinical malaria, and it is
geographically widespread. _P. vivax_ had been considered as a "benign" malaria for a long time, but recent studies have shown that it can cause severe consequences including
death2. While existing anti-malarial control measures, such as insecticide-treated nets, rapid diagnosis, and antimalarial drugs, have reduced malaria cases and deaths dramatically in the
last two decades, the numbers are similar between 2014 and 20181. Therefore, in addition to expanding applications of current control measures, efforts must to be made to develop new tools,
such as transmission-blocking vaccines (TBVs)3. For the development of TBVs as well as transmission-blocking drugs (TBD), an assay which can evaluate reduction or complete blocking of
parasite growth in mosquitoes is indispensable. The standard membrane-feeding assay (SMFA) or direct membrane-feeding assay (DMFA) have been used widely for the development of
transmission-blocking interventions against both _P. falciparum_ and _P. vivax_3,4,5,6,7,8,9. The membrane-feeding assays have been also utilized to evaluate transmission-reducing immunity
induced by natural malaria infections10,11 and to determine vector competence in transgenic mosquitoes12, which is one of the alternative strategies to reduce malaria transmission in the
field. SMFA is conducted with cultured sexual-stage parasites (gametocytes), while DMFA uses blood samples from _P. falciparum_- or _P. vivax_-infected patients as the source of gametocytes.
Since there is no reliable methodology to transport gametocytes for a long distance without losing their infectivity, usually DMFA is performed at (or near) the field site where the blood
samples are collected. It is more challenging to control the quality of assays in the field site compared to an assay performed at fully equipped laboratories with updated infrastructure. In
addition, different blood samples have different density and/or maturity of gametocytes, and different male-and-female gametocyte ratios, all of which affect infectivity to mosquitoes13.
SMFA can be performed in more controlled conditions, but on the opposite side, it is difficult to mimic the complexity of parasites seen in the field. For _P. falciparum_, the great majority
of SMFAs have been done with NF54 (or the clones from NF54, e.g., 3D7) strains of parasites, and only a limited number of other strains are applicable for SMFA, because strains which can
consistently generate high enough gametocytemia in in vitro cultures are rare. For _P. vivax_, it is impossible to prepare cultured gametocytes, so DMFA is the only option at this moment. At
the early stage of TBV and TBD development, it is almost routine to compare biological activity of different candidates (e.g., different antigens, adjuvants, drugs); thus determination of
the error range for the measured activity, usually expressed as % inhibition in oocyst density (also called "transmission-reducing activity", or "%TRA") or % inhibition
in prevalence of infected mosquitoes (also referred to as "transmission-blocking activity", or "%TBA"), is essential. However, in many studies, observed % inhibition
values were reported without proper error ranges (e.g., 95% confidence interval, 95% CI) and a non-parametric statistical test (e.g., a Mann–Whitney test) was used in many cases to compare
different groups. While the approach is reasonable to compare the candidates tested within the same assay, it is almost impossible to properly compare different candidates tested in
different feeding assays (even within a single paper) when the assays show different levels of mean oocyst in the control groups. Early studies in the 1990’s have shown that distribution of
oocysts for rodent (_P. berghei_), chicken (_P. gallinaceum_), and human (_P. falciparum_ and _P. vivax_) malarias in various _Anopheles_ mosquitoes can be better explained by a negative
binomial (NB) model than a normal (Poisson) distribution14,15; recent studies have demonstrated that a zero-inflated negative binomial (ZINB) model is better than a NB model for SMFA with
_P. falciparum_ (PfSMFA)16,17 and _P. berghei_16. In addition to the ZINB models, a beta-binomial model has been used in another study for PfSMFA18. These mathematical models allow the
calculation of uncertainty around the measured %TRA (e.g., 95% CI) and support a rational comparison of multiple candidates in SMFA. A recent study for DMFA with _P. vivax_ (PvDMFA) again
demonstrated that the oocyst data deviated from a Poissonian prediction19. However, no mathematical model has been published to interpret DMFA data. Our basic hypothesis was that a ZINB
model, which has been shown to be useful for PfSMFA, could be universally utilized to analyze the results of any membrane-feeding assay. If this hypothesis is true, the ZINB model can
support better designing, reporting and interpreting of all membrane-feeding assays. To this end, in this study, we compared two distinct membrane-feeding assays with human malaria
parasites, SMFA with _P. falciparum_ NF54 strain using _Anopheles stephensi_ mosquitoes (the details of model fitting have been published previously17,20) and DMFA with _P. vivax_ using _An.
dirus_ mosquitoes (new data collected from 22,236 mosquitoes in 96 independent assays). While there were significant differences in the best-fit parameters between the two assays, PvDMFA
data could be explained reasonably well by a ZINB model, as is PfSMFA. A brief explanation of each parameter of the ZINB model and abbreviations used in this manuscript are summarized in
Table 1. We then evaluated the impact of each parameter on the accuracy of % inhibition estimates. Lastly, we simulated how modifications to the assay design (e.g., number of mosquitoes
examined per group, performing repeat PvDMFA for the same samples) change the error range of %inhibition estimates. The simulation results will not only support designing new DMFA and SMFA
experiments, but also help rational comparisons for transmission-reducing activities among different candidates when the proper error of measurement is not reported. RESULTS AND DISCUSSION
ESTABLISHMENT OF PVDMFA-SPECIFIC MODEL _Plasmodium falciparum_ SMFA (PfSMFA) results have been shown to be explained well with a zero-inflated negative binomial (ZINB) model16,17. To assess
whether a similar ZINB model is applicable for _P. vivax_ DMFA (PvDMFA), the correlations between mean and standard deviation (SD) of oocyst intensity, and between mean oocyst intensity and
prevalence of infected mosquitoes were evaluated first. For the analysis, PvDMFA data from 96 independent assays with 22,236 mosquitoes tested in a total of 1,022 "Container of
Mosquitoes" (COM) were utilized. COM means a group of mosquitoes which were housed in the same container and were fed the same blood-test (or control) antibody mixture. The average
number of mosquitoes per COM was 21.8. While more PvDMFA data were available, we only used data with at least one infected mosquito in each COM (if there is no infected mosquito, there is no
difference between PvDMFA and PfSMFA, as mean, SD and prevalence are all zero). The original PvDMFA data are seen in Supplementary Table S1. The PvDMFA were performed with _P. vivax_
gametocytes collected from _P. vivax_ patients and laboratory-reared _An. dirus_ mosquitoes, while the PfSMFA were all performed with cultured _P. falciparum_ (NF54) gametocytes and
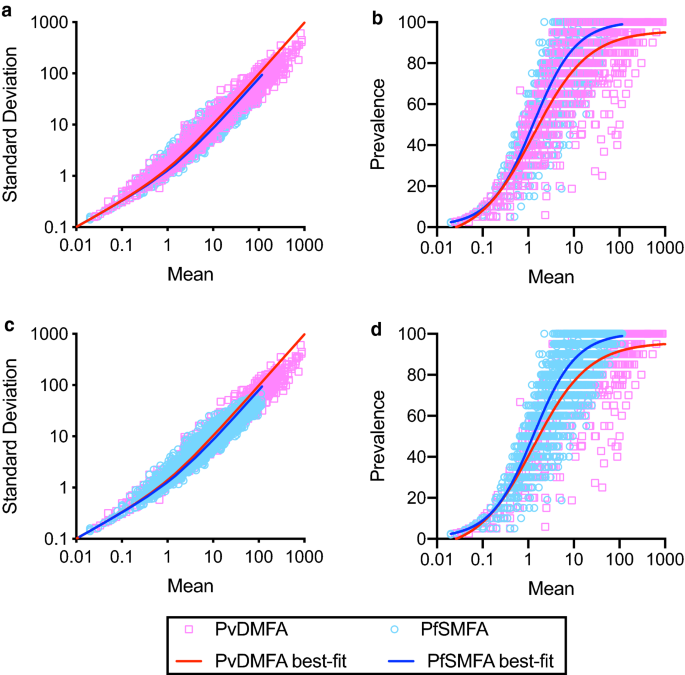
laboratory-reared _An. stephensi_ mosquitoes. Although there were obvious differences in methodologies, as shown in Fig. 1a,c, the best-fit curves (calculated by a negative binomial fit) for
mean-SD associations in two different assays overlapped almost perfectly. While the negative binomial fit line for the PvDMFA data appears to be too high at higher mean oocysts (> 100
oocysts), still the model fits the data reasonably well (R2 = 0.951). For mean-prevalence correlations (Fig. 1b,d), the best-fit curves (by a sigmoid curve fit) were similar overall, but the
PvDMFA curve shifted slightly to the right. Strong, and similar associations of mean-SD and mean-prevalence were reported in SMFA with rodent malaria14,16 and DMFA with _P. falciparum_
parasites21,22 tested in multiple anopheline mosquitoes. The previous and current studies indicate that ZINB models could be usefully used to analyze feeding assays performed with multiple
strains of parasites and mosquitoes, while each parameter value needs to be optimized depending on assays. The similarity (mean-SD and mean-prevalence associations) in different assays
suggest that a beta binomial (BB) model could also be used for PvDMFA data as shown in a recent paper with PfSMFA18. Both the beta-binomial and negative-binomial models are ways to handle
overdispersed count data. The beta-binomial model is for proportions and it requires a maximum count to be declared, while the negative binomial model addresses counts and does not require
the maximum count declaration. The "zero-inflated" negative binomial has an additional parameter that allows estimating the proportion of mosquitoes that would never develop
oocysts under any circumstance. It is mathematically easy to compare ZINB and NB models, because the NB model is nested within the ZINB model. However, since BB and ZINB models are
completely different ones, it is difficult to examine which model is significantly better than the other with the data used in this analysis, therefore, we will only discuss ZINB models in
this manuscript. A PvDMFA-specific ZINB model (R2 = 0.956) was generated using results of PvDMFA where normal human AB + serum was tested (a total of 2,226 mosquitoes tested in 105 COMs by
63 independent assays), and the data allowed estimation of feed-to-feed and COM-to-COM variations (true %TRA of all normal serum COMs was considered to be zero). The details of PfSMFA data
and analysis have been published before17,20,23. As expected from Fig. 1a,c, the negative binomial dispersion parameter θ are similar for PvDMFA and PfSMFA (Table 2, Model #1 vs. #4). On the
other hand, zero-inflation parameter π, and the random effects of feed (Vf) and COM (Vc) are higher in PvDMFA compared to PfSMFA. For example, a model with π = 0.179 means that we expect
that regardless of the amount of antibody present (or absent), at least 17.9% of the mosquitoes will not produce any oocysts. Higher Vf and Vc indicate that larger feed-to-feed and
COM-to-COM variations, respectively, are predicted in PvDMFA than in PfSMFA when the same sample is tested in the same number of repeat feeds or multiple COMs (in a feed). We next assessed
the impact of each of the best-fit parameters in Model #1 (PvDMFA, red in Fig. 2) and #4 (PfSMFA, blue in Fig. 2) on %TRA estimations by simulations. In each iteration of the simulation, the
best estimate %TRA and the 95% confidence interval (95% CI) for a test was calculated. From the 95% CI values, 95% CI length was determined as an indicator of the accuracy. For example,
when a sample is tested by a feeding assay, and the high end of 95% CI (H95% CI) is estimated as 85% TRA, and the low end of 95% CI (L95% CI) as 60% TRA, the 95% CI length is calculated as
85–60 = 25. The median value of the 95% CI length (y-axis in Fig. 2) was obtained from 10,000 iterations for each scenario. PvDMFA data showed bigger 95% CI length than that in PfSMFA (Fig.
2), indicating more uncertainty in the %TRA estimates. In addition to the accuracy of %TRA estimate (how large or small was the 95% CI length), it is equally important to assess whether an
observed inhibition is significant or not. Since the highest end of %TRA is 100% by definition (i.e., test COM has no oocysts), H95% CI cannot exceed 100% as well. Therefore, if 95% CI
length of a test sample becomes larger than 100 (above the dotted horizontal line in Fig. 2), the L95% CI should be lower than 0% TRA, i.e., the observed %TRA is not significantly different
from no inhibition. For example, the median 95% CI length of Model #1 at 80% TRA was 129. The result suggests that when a sample with true 80% TRA activity is tested in multiple PvDMFA
assays (when the assays are performed as the simulated assay condition), at least in half of the assays, the observed inhibitions are judged as insignificant (i.e., L95% CI is lower than
zero). The actual proportion of feeding experiments which show significant inhibitions is shown in Supplementary Fig. S1. Further simulations were performed to determine how the 3 parameters
(π, Vf and Vc), which differed between PvDMFA and PfSMFA, effect the accuracy by switching each parameter of Model #1 to the corresponding one in Model #4. As clearly shown in Fig. 2, when
the Vc parameter (COM-to-COM variation within a feed) from the PvDMFA model was replaced with that in PfSMFA model, the median 95% CI length reduced significantly, almost similar to the 95%
CI length seen in the PfSMFA model (Model #4). On the other hand, replacing π or Vf values from PvDMFA model to those in PfSMFA model had minimum impact on the 95% CI length. The results
clearly showed that higher Vc in PvDMFA model had the most influential effect on the uncertainty (i.e., bigger 95% CI length) in %TRA estimates, not higher π or Vf. In other words, if one
modifies the assay, by which Vc can be reduced, the accuracy of %TRA estimates should be increased dramatically. Previous studies with PfSMFA16,20 have shown that higher mean control oocysts
(mo-contl) yields more accurate estimates of %TRA (if all the other conditions are the same). Therefore, modified models were generated by excluding feeding data with lower mo-contl (Model
#2, 3, 5 and 6 in Table 2). In the analysis, two different cut-off values of exclusion were evaluated; 1 (i.e., excluded all COMs with < 1 mo-contl) and 4 (i.e., excluded COMs with < 4
mo-contl). In both PvDMFA and PfSMFA, Vf and Vc parameters went down gradually by increasing the cut-off value of exclusion from zero (no exclusion, Models #1 and 4) to 1 (Models #2 and 5)
to 4 (Models # 3 and 6). The advantage of those modified models was assessed by additional simulations (Fig. 3). When feeding data with less than 4 mo-contl were excluded from the analysis
(Model #3), the median 95% CI lengths became close to PfSMFA data. In the Model #3, the median 95%CI length line crossed y = 100 (black dotted line in Fig. 3) at ~ 60% TRA level of activity,
instead of ~ 85% TRA in Model #1. The proportion of feeds which demonstrate significant inhibitions is shown in Supplementary Fig. S2. When the cut-off value was increased more (e.g., 5 or
10, instead of 4), Vc (and also Vf and π) was further decreased, i.e., it increased the accuracy of %TRA estimates, as expected (data not shown). However, at this moment, there is no
reliable way to keep mo-contl at a certain level (e.g., 4 or higher) in any given feeding experiment for DMFA and SMFA. Therefore, if the exclusion threshold is set to be higher, it reduces
the throughput of assay (i.e., discard more DMFA or SMFA results when the control group does not reach to the determined threshold level of mean oocyst density). The best balance of
throughput (reduce the threshold) and accuracy of %TRA estimates (increase the threshold) should be determined based on the usage of the assay. The results emphasize the importance of
reporting %TRA estimates with the error ranges (e.g., 95% CI), not only presenting observed %TRA values, especially when the error range of %TRA estimates is large (e.g., assay with high
COM-to-COM variation, using a lower exclusion threshold). In the remainder of this article, the threshold of 4 was selected, as we thought it was a practically acceptable criterion. In other
words, Model #3 was used for all following simulations unless specified, and any real PvDMFA data which came from feeds with < 4 mo-contl were excluded from analysis. TWO DIFFERENT
READOUTS: TRA AND TBA Two different readouts have been used to express SMFA and DMFA results; %inhibition in oocyst intensity (%TRA) and %inhibition in prevalence of infected mosquitoes
(%TBA). In the case of PfSMFA, it has been shown that %TBA results cannot be interpreted well without mo-contl data, while %TRA is independent of the readout from the mo-contl (at least at
the level of mo-contl evaluated in the studies)16,20. More importantly, when model-based %TBA was calculated from observed %TRA and observed mo-contl using the ZINB model, there was a strong
concordance between the observed %TBA and model-based %TBA20. The strong concordance has proved that %TBA and %TRA are not independent readouts, and %TBA may be determined by %TRA, mo-contl
and the ZINB model. To assess whether there is the same interaction between %TRA, mo-contl and %TBA in PvDMFA, observed %TBA and model-based %TBA were compared (Fig. 4). As shown in Fig. 4a
(mo-contl ranged from 4.3 to 8.2) and Fig. 4b (mo-contl ranged from 8.2 to 18.3) when mo-contl were lower, there were strong concordances between observed %TBA and model-based %TBA as seen
in PfSMFA20: RMAC = 0.80 and 0.75 for 1st quarter (Q) and 2nd Q, respectively (p < 0.0001 for both). The results indicate again that %TBA is a correlate of mo-contl and %TRA, and that
%TBA and %TRA are not independent measurements, even for PvDMFA. However, when mo-contl became higher (3rd Q, mo-contl ranged from 18.3 to 64.3; Fig. 4c,e), the RMAC value went down (RMAC =
0.46, p < 0.001), and low and insignificant concordance was observed at the highest mo-contl group (4th Q, mo-contl ranged from 64.3 to 303; Fig. 4d,e, RMAC = − 0.03, p = 0.73). In
theory, %TBA converges on zero when mo-contl becomes bigger when the same sample is tested. For example, if a control group has 300 oocysts on average, even if a test antibody reduces the
average number of oocysts by 90% (i.e., 90% TRA), the average oocysts of the test group is 30, and the proportion of no-oocyst mosquitoes should be very minor (i.e., 0% TBA). As predicted,
the majority of model-based %TBA values were ~ 0% in Fig. 4d, on the other hand, observed %TBA varied from − 36 to 77%. Why was there a discrepancy between observed %TRA and model-based %TBA
values at higher mo-contl data set? As shown in Table 2, zero-inflation parameter π is bigger in PvDMFA than in PfSMFA. In other words, regardless of antibody activity (or no activity),
more mosquitoes in a COM are likely to have zero oocysts in PvDMFA. Therefore, in PvDMFA, even when average oocyst number of a control COM is high, such as 4th Q in Fig. 4, a larger
proportion of mosquitoes (both in control and test COMs) display zero oocysts by chance, not due to the transmission-blocking activity of test sample, compared to PfSMFA. While the effect of
such zero-oocyst-mosquitoes on mean oocysts (which is used to calculate %TRA) is relatively smaller in a higher mo-contl feed, the zero-oocyst mosquitoes directly change observed %TBA
values. Therefore, we hypothesized that higher π parameter in PvDMFA led to larger errors in observed %TBA values. To test the hypothesis, simulations were performed at various π levels.
Four different levels of π parameter (no zero-inflation, π = 0; same as PfSMFA, π = 0.06; same as PvDMFA, π = 0.16; and doubling for PvDMFA, π = 0.32) were tested with three different levels
of %TRA samples (no inhibition, %TRA = 0; moderate inhibition, %TRA = 60, and strong inhibition, %TRA = 90). The other 4 parameters (i.e., mo-contl, θ, Vf and Vc) were kept as in Model #3.
In all 3 levels of %TRA, %TBA values diverged more as π became bigger (Fig. 5a), indicating more uncertainty in %TBA estimates. Next, the proportion of the data set which were categorized
into one of four areas was evaluated (Fig. 5b). Theoretically, all data should be categorized into “Area 1 (%TRA ≥ 0 & %TBA ≥ 0)” (when inhibitory samples are tested) or "Area 3
(%TRA < 0 & %TBA \(\le \) 0)" (when enhancing samples are tested), but due to the error of assay, some data fell into "Area 2 (%TRA < 0 & %TBA > 0)" and
"Area 4 (%TRA > 0 & %TBA < 0)". When 0% TRA sample was tested, %TRA and %TBA values could be positive or negative just by chance, and the simulation results were
scattered into all 4 areas as predicted (but a majority of the data were within Areas 1 and 3). When an inhibitory sample was tested (%TRA = 60 or 90 in this case), in theory (i.e., if there
is no error in the assay) all data should be classified into Area 1, and indeed, the largest proportion of data set in all scenarios was grouped into Area 1. However, a considerable number
of data sets fell into Area 4 even with π = 0.06 (same level as PfSMFA), and the proportion further increased as π parameter was larger (Fig. 5b). When a DMFA (or SMFA) result happens to
fall into Area 3 or 4 classes, an investigator might conclude the result as “enhancement” of infection (prevalence) when they use observed %TBA readouts without considering the error of the
assay. Similarly, Area 2 or 3 data could be considered as “enhancement” of oocyst density. However, the simulation showed that the proportion of Area 2 or 3 results was much smaller than
that in Area 4 when an inhibitory sample was tested (true %TRA of 60 or 90). These simulations strongly suggest that observed %TBA could be very misleading, especially for an assay with
higher π. If the issue of using observed %TBA as a PvDMFA readout is the random error in measurement for prevalence of infected mosquitoes, one might think that the problem could be overcome
by examining more mosquitoes per COM, not 20 mosquitoes per COM as in Fig. 5. Therefore, the impact of number of mosquitoes on %TBA readout was next assessed with the fixed π parameter
(0.16). As expected, %TBA values diverged less when more mosquitoes were dissected (Fig. 6a). However, even when 2,000 mosquitoes per COM were examined, ~ 20% and ~ 5% of data fell into Area
4 at 60% or 90% TRA, respectively (Fig. 6b). Since dissecting 2,000 (or more) mosquitoes per COM is practically impossible, it is more realistic to use %TRA as the main readout of PvDMFA.
Of note, as shown in previous studies with PfSMFA16,20, observed %TRA values are more reliable at higher mo-contl, but opposite for %TBA; observed %TBA is less reliable at higher mo-contl
(Fig. 4). IMPACT OF ASSAY MODIFICATIONS ON ACCURACY OF %TRA ESTIMATES Lastly, the impact of assay modifications on %TRA estimates was evaluated. At 4 levels of mo-contl (minimum number
recommended, 4; 25th percentile of PvDMFA data which were used to generate Model #3, 10.4; 50th percentile, 22.1; and 75th percentile, 95.6), 3 levels of %TRA (60, 80 and 90%) samples were
examined in 9 different assay conditions (number of mosquitoes dissected per COM was 20, 40 or 60; the same test sample was evaluated in 1, 2 or 3 independent feeding experiments). Figure 7
shows median of L95% CI and H95% CI of %TRA estimate from 10,000 iterations (per scenario). For example, when a test sample with 60% TRA activity was examined in a single feed and n = 20 per
COM (20 mosquitoes were dissected in the control group, and another 20 mosquitoes for test group), and the control group in the feed showed 4 oocysts on average (mo-contl = 4), median L95%
CI was calculated as − 33, and H95% CI was 89 (top left panel in Fig. 7). Since the L95% CI was lower than zero (no inhibition), it indicates that a sample with 60% TRA activity is unlikely
to show a significant inhibition in at least > 50% of feeds. As predicted, more mosquitoes, more feeds, and higher mo-contl could increase the accuracy of the %TRA estimate (shorter bars
in Fig. 7). When mo-contl is very low, such as < 4 oocysts on average, a small change in the mo-contl substantially affects the accuracy of %TRA estimate (Fig. 3 and previous
studies16,17). However, the 95% CI lengths in mo-contl = 10.4 scenarios were not so different from those in mo-contl = 4 scenarios when the same sample was tested with the same test
condition. Similar minor differences were observed for mo-contl = 10.4 vs. 22.1, or 22.1 vs. 95.6. The results suggest that it is not so critical to obtain higher mo-contl once it reaches a
certain level. In these simulations, up to 60 mosquitoes per COM, and up to 3 repeat feeds were evaluated. For the laboratories which routinely perform PvDMFA, such modifications are
considered as “doable” options, and some laboratories might do more. However, those require more time and resources, and a benefit of the modification (i.e., how much of 95% CI length can be
narrowed down) is reduced as %TRA level goes up. For example, when a 60% TRA sample was tested at mo-contl = 4 (top left panel in Fig. 7), the 95% CI range was − 33 to 89% with n = 20 × 1
feed, and 19 to 84% with n = 60 × 3 feeds. By dissecting 9 times more mosquitoes, the median L95% CI became > 0 (i.e., now more than 50% of the time, one can call that the observed
inhibition is significantly better than no inhibition), and the 95% CI length turned into 65 (= 84 − 19; n = 60 × 3 feeds) from 122 (= 98 − (− 33); n = 20 × 1 feed). On the other hand, with
the same modification, the reduction in 95%CI length was only 15 (= 34 − 19) when a 90% TRA sample was tested (bottom left panel in Fig. 7). Depending on the target level of inhibitory
activity which the investigators want to detect and desired accuracy in %TRA estimates, the methodology of PvDMFA (e.g., number of mosquitoes, repeat feeds) should be optimized, and
simulations, such as shown in Fig. 7, will support the design of the assay. Not only for this purpose, the simulation results also help a proper interpretation of an observed %TRA value when
the error range is not specified in the original report. As shown in Fig. 3, the median 95% CI length in PfSMFA and PvDMFA are reasonably close when assays with fewer mean control oocysts
(i.e., < 4 oocysts on average in the simulations) are excluded. It suggests that investigators could use the results shown in Fig. 7 (or similar simulations) to estimate an error range of
an observed %TRA either from SMFA or DMFA, or either with _P. falciparum_ or _P. vivax_. To the best of our knowledge, this is the first study which evaluates the model-fit and the impact
of each parameter for DMFA data. Since DMFA uses patient blood as the source of gametocytes, and each blood sample contains different parasite mixtures, it is predicted that PvDMFA would
show higher assay-to-assay variation (Vf). However, a mechanism of higher π and Vc in PvDMFA was not investigated in this study, because many factors could contribute to the differences in
the parameters (e.g., _P. falciparum_ vs. _P. vivax_, _An. stephensi_ vs. _An. dirus_, SMFA vs. DMFA). To uncover the mechanism(s), a much larger set of data (e.g., PfSMFA performed with
_An. dirus_, PvDMFA with _An. stephensi_) are likely to be required. As shown in Fig. 2, Vc parameter has a substantial effect on accuracy of %TRA estimates, therefore, it is ideal to
determine at least the Vc parameter for each specific DMFA (or SMFA) condition in each laboratory. A second point which is not evaluated in this study is applicability of a ZINB model for
other transmission-blocking interventions. All of the PfSMFA and PvDMFA data were collected during TBV development; i.e., to evaluate biological activity of vaccine induced antibodies. These
strong associations between mean-SD and between mean-prevalence were similarly reported in studies which included data from transmission-blocking drugs16 and transmission-blocking
antimicrobial peptides24. Therefore, we think it is possible to apply a similar ZINB model to evaluate other transmission-blocking interventions, other than TBVs (although each parameter
needs to be reevaluated). However, if an intervention has a totally different mechanism of action (e.g., killing only oocyst-stages of parasites, instead of targeting
gametocyte/gamete/zygote/ookinete parasites), a different mathematical model may fit better than a ZINB model. This study also shows multiple commonalities between PvDMFA and PfSMFA, despite
the fact that there are significantly differences in the assays in terms of parasite species (_P. vivax_ and _P. falciparum_), source of gametocytes (patients’ blood and cultured
gametocytes), mosquito species (_An. dirus_ and _An. stephensi_), and geography (Thailand and United States). This study strongly suggests that the multiple commonalities found in this study
can be applied for any other membrane-feeding assays, at least assays with human malaria parasites. Those include; (1) both data types can be explained by a zero-inflated negative binomial
model, (2) the negative binomial dispersion parameter θ was similar, (3) excluding feeds with lower control oocysts (mo-contl) increases accuracy of %TRA estimates, and (4) %TBA is
determined by %TRA and mo-contl, not an independent readout. In addition, when one wants to optimize or standardize DMFA or SMFA, not only control oocysts (mo-contl), but also COM-to-COM
variation (Vc) need to be evaluated during the process. Based on the common ground between DMFA and SMFA, it is recommended; (1) to report %TRA with a proper error range (e.g., 95% CI), and
(2) not to use observed %TBA as a main readout, because %TRA gives fairer comparisons of feeding results from different experiments and/or different investigators. Furthermore, a recent
study by Churcher et al. has demonstrated that the intensity of a mosquito infection is critically important to the success of transmission25. The results suggest that %TRA readout could be
superior than %TBA even from a biological point of view. Nevertheless, if for any reason it is preferred to use %TBA readout, we recommend using “standardized %TBA”17 rather than observed
%TBA. Under the ZINB model %TBA is reasonably estimated from observed %TRA and mo-contl in PvDMFA (Fig. 4a,b in this paper) and in PfSMFA (as published before20). The “standardized %TBA” is
a model-based %TBA calculated from observed %TRA and a giving mo-contl level, which an investigator can specify any value. This study should support development of future
transmission-blocking vaccines, and likely transmission-blocking drugs as well, to understand the pros and cons of DMFA and SMFA, to help better designing and reporting for future DMFA and
SMFA experiments, and to aid rational comparisons of different candidates by properly interpreting the DMFA and SMFA results even when an error range of observed %TRA value is not described
in the original report. METHODS DIRECT MEMBRANE FEEDING ASSAY WITH _P. VIVAX_ Patients visiting malaria clinics in north-west Thailand were microscopically examined for their malaria
infections. When diagnosed with _P. vivax_ infection and ≥ 15 years old, they were invited to donate their blood for PvDMFA. The protocol for blood collection from the patients was approved
by Ministry of Public Health Ethical Committee, Bangkok, Thailand (protocol # WRAIR1308), and written informed consent was obtained from all volunteers. The details of PvDMFA methodology
have been published elsewhere26. In brief less than two hours after collection, heparinized blood was aliquoted at 350 μL per tube before being centrifuged, and the plasma was removed. The
packed erythrocytes were washed with incomplete RPMI medium to reduce the impact of patient plasmas on infectivity of gametocytes. Each tube with packed erythrocytes mixed with 180 μL of
test antibodies (either sera or purified IgG samples) and a pool of normal human AB + serum was immediately placed in feeding apparatuses and offered to _Anopheles dirus_ mosquitoes. The
mosquitoes were allowed to feed on infected blood for 30 min. The blood samples were kept at 37–38 °C as much as possible using a temperature-control container during blood transportation
and a waterbath during blood processing and feeding. After removal of unfed mosquitoes, remaining mosquitoes were kept at the insectary at 28 °C for 7 to 9 days before dissection. The
oocysts in the midguts were examined and counted by microscopy. All methods were performed in accordance with the relevant guidelines and regulations. The original oocyst count in each
individual mosquito is seen in Supplementary Table S1. MODEL BUILDING AND STATISTICAL ANALYSIS The ZINB model displayed in Table 2 was parameterized like the representative count model as
described before17 by R with the package glmmTMB27. For Figs. 2, 3, and 7, data was simulated using the described parameters in different scenarios. In each iteration for each scenario of
simulation, the 95% CIs were calculated using the ZINB-RE Sim 1 method as described in17. For Fig. 4, model-based %TBA was calculated as described in17 and the random marginal agreement
coefficients (RMAC) analysis was performed28. RMAC is an agreement measure that is on the same scale as correlations (− 1 to 1), with 0 being agreement observed is the same as chance, and 1
being perfect agreement. The RMAC code is available upon request. The number of iterations in each scenario of simulation was described in the corresponding figure legend. DATA AVAILABILITY
All original PvDMFA data are included in Supplementary Table S1. REFERENCES * World Health Organization. World malaria report 2019.
https://www.who.int/publications-detail/world-malaria-report-2019. Accessed 29 March 2020 (2019). * Baird, J. K. Evidence and implications of mortality associated with acute _Plasmodium
vivax_ malaria. _Clin. Microbiol. Rev._ 26, 36–57 (2013). Article Google Scholar * Miura, K. _et al._ Malaria transmission-blocking vaccines: Wheat germ cell-free technology can accelerate
vaccine development. _Expert Rev. Vaccines_ 18, 1017–1027 (2019). Article CAS Google Scholar * Smit, M. R. _et al._ Human direct skin feeding versus membrane feeding to assess the
mosquitocidal efficacy of high-dose Ivermectin (IVERMAL Trial). _Clin. Infect. Dis._ 69, 1112–1119 (2018). Article Google Scholar * McCaffery, J. N. _et al._ A multi-stage _Plasmodium
vivax_ malaria vaccine candidate able to induce long-lived antibody responses against blood stage parasites and robust transmission-blocking activity. _Front. Cell Infect. Microbiol._ 9, 135
(2019). Article CAS Google Scholar * Colmenarejo, G. _et al._ Predicting transmission blocking potential of anti-malarial compounds in the mosquito feeding assay using _Plasmodium
falciparum_ male gamete inhibition assay. _Sci. Rep._ 8, 7764 (2018). Article ADS Google Scholar * Vos, M. W. _et al._ A semi-automated luminescence based standard membrane feeding assay
identifies novel small molecules that inhibit transmission of malaria parasites by mosquitoes. _Sci. Rep._ 5, 18704 (2015). Article ADS CAS Google Scholar * Arevalo-Herrera, M. _et al._
Recombinant Pvs48/45 antigen expressed in _E. coli_ generates antibodies that block malaria transmission in _Anopheles albimanus_ mosquitoes. _PLoS ONE_ 10, e0119335 (2015). Article Google
Scholar * Tachibana, M. _et al._ _Plasmodium vivax_ gametocyte proteins, Pvs48/45 and Pvs47, induce transmission-reducing antibodies by DNA immunization. _Vaccine_ 33, 1901–1908 (2015).
Article CAS Google Scholar * Stone, W. J. R. _et al._ Unravelling the immune signature of _Plasmodium falciparum_ transmission-reducing immunity. _Nat. Commun._ 9, 558 (2018). Article
ADS Google Scholar * Ouedraogo, A. L. _et al._ Modeling the impact of _Plasmodium falciparum_ sexual stage immunity on the composition and dynamics of the human infectious reservoir for
malaria in natural settings. _PLoS Pathog._ 14, e1007034 (2018). Article Google Scholar * Mumford, J. D. _et al._ _Plasmodium falciparum_ (Haemosporodia: Plasmodiidae) and O’nyong-nyong
virus development in a transgenic _Anopheles gambiae_ (Diptera: Culicidae) strain. _J. Med. Entomol._ 56, 936–941 (2019). Article Google Scholar * Bousema, T., Churcher, T. S., Morlais, I.
& Dinglasan, R. R. Can field-based mosquito feeding assays be used for evaluating transmission-blocking interventions?. _Trends Parasitol._ 29, 53–59 (2013). Article Google Scholar *
Medley, G. F. _et al._ Heterogeneity in patterns of malarial oocyst infections in the mosquito vector. _Parasitology_ 106(Pt 5), 441–449 (1993). Article Google Scholar * Billingsley, P.
F., Medley, G. F., Charlwood, D. & Sinden, R. E. Relationship between prevalence and intensity of _Plasmodium falciparum_ infection in natural populations of _Anopheles_ mosquitoes. _Am.
J. Trop. Med. Hyg._ 51, 260–270 (1994). Article CAS Google Scholar * Churcher, T. S. _et al._ Measuring the blockade of malaria transmission—An analysis of the standard membrane feeding
assay. _Int. J. Parasitol._ 42, 1037–1044 (2012). Article Google Scholar * Swihart, B. J., Fay, M. P. & Miura, K. Statistical methods for standard membrane-feeding assays to measure
transmission blocking or reducing activity in malaria. _J. Am. Stat. Assoc._ 113, 534–545 (2018). Article MathSciNet CAS Google Scholar * Dechering, K. J. _et al._ Modelling mosquito
infection at natural parasite densities identifies drugs targeting EF2, PI4K or ATP4 as key candidates for interrupting malaria transmission. _Sci. Rep._ 7, 17680 (2017). Article ADS
Google Scholar * Kiattibutr, K. _et al._ Infectivity of symptomatic and asymptomatic _Plasmodium vivax_ infections to a Southeast Asian vector, _Anopheles dirus_. _Int. J. Parasitol._ 47,
163–170 (2017). Article Google Scholar * Miura, K. _et al._ Transmission-blocking activity is determined by transmission-reducing activity and number of control oocysts in _Plasmodium
falciparum_ standard membrane-feeding assay. _Vaccine_ 34, 4145–4151 (2016). Article Google Scholar * Goncalves, B. P. _et al._ Examining the human infectious reservoir for _Plasmodium
falciparum_ malaria in areas of differing transmission intensity. _Nat. Commun._ 8, 1133 (2017). Article ADS Google Scholar * Bompard, A. _et al._ Evaluation of two lead malaria
transmission blocking vaccine candidate antibodies in natural parasite-vector combinations. _Sci. Rep._ 7, 6766 (2017). Article ADS Google Scholar * Miura, K. _et al._ Qualification of
standard membrane-feeding assay with _Plasmodium falciparum_ malaria and potential improvements for future assays. _PLoS ONE_ 8, e57909 (2013). Article ADS CAS Google Scholar *
Habtewold, T. _et al._ Streamlined SMFA and mosquito dark-feeding regime significantly improve malaria transmission-blocking assay robustness and sensitivity. _Malar. J._ 18, 24 (2019).
Article Google Scholar * Churcher, T. S. _et al._ Probability of transmission of malaria from mosquito to human is regulated by mosquito parasite density in naive and vaccinated hosts.
_PLoS Pathog._ 13, e1006108 (2017). Article Google Scholar * Sattabongkot, J., Kumpitak, C. & Kiattibutr, K. Membrane feeding assay to determine the infectiousness of _Plasmodium
vivax_ gametocytes. _Methods Mol. Biol._ 1325, 93–99 (2015). Article CAS Google Scholar * Brooks, M. E. _et al._ glmmTMB balances speed and flexibility among packages for zero-inflated
generalized linear mixed modeling. _R J._ 9, 378–400 (2017). Article Google Scholar * Fay, M. P. Random marginal agreement coefficients: Rethinking the adjustment for chance when measuring
agreement. _Biostatistics_ 6, 171–180 (2005). Article Google Scholar Download references ACKNOWLEDGEMENTS This study was supported by the Intramural Research Program of the National
Institute of Allergy and Infectious Diseases, NIH. AUTHOR INFORMATION Author notes * These authors contributed equally: Kazutoyo Miura and Bruce J. Swihart. AUTHORS AND AFFILIATIONS *
Laboratory of Malaria and Vector Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health, 12735 Twinbrook Parkway, Rockville, MD, 20852, USA Kazutoyo
Miura & Carole A. Long * Biostatistics Research Branch, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Rockville, MD, USA Bruce J. Swihart &
Michael P. Fay * Mahidol Vivax Research Unit, Faculty of Tropical Medicine, Mahidol University, Bangkok, Thailand Chalermpon Kumpitak, Kirakorn Kiattibutr & Jetsumon Sattabongkot Authors
* Kazutoyo Miura View author publications You can also search for this author inPubMed Google Scholar * Bruce J. Swihart View author publications You can also search for this author
inPubMed Google Scholar * Michael P. Fay View author publications You can also search for this author inPubMed Google Scholar * Chalermpon Kumpitak View author publications You can also
search for this author inPubMed Google Scholar * Kirakorn Kiattibutr View author publications You can also search for this author inPubMed Google Scholar * Jetsumon Sattabongkot View author
publications You can also search for this author inPubMed Google Scholar * Carole A. Long View author publications You can also search for this author inPubMed Google Scholar CONTRIBUTIONS
K.M. and B.J.S.: conception and design, analysis and interpretation of data, and draft the paper. C.K. and K.K.: acquisition of data, and revision of article. J.S.: conception and design,
acquisition of data, and revision of article. M.P.F.: conception and design, analysis and interpretation of data, and revision of article. C.A.L.: conception and design, interpretation of
data, revision of article. All authors read and approved the final manuscript. CORRESPONDING AUTHOR Correspondence to Kazutoyo Miura. ETHICS DECLARATIONS COMPETING INTERESTS The authors
declare no competing interests. ADDITIONAL INFORMATION PUBLISHER'S NOTE Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional
affiliations. SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES. SUPPLEMENTARY TABLE S1. RIGHTS AND PERMISSIONS OPEN ACCESS This article is licensed under a Creative Commons Attribution 4.0
International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the
source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative
Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by
statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit
http://creativecommons.org/licenses/by/4.0/. Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Miura, K., Swihart, B.J., Fay, M.P. _et al._ Evaluation and modeling of direct
membrane-feeding assay with _Plasmodium vivax_ to support development of transmission blocking vaccines. _Sci Rep_ 10, 12569 (2020). https://doi.org/10.1038/s41598-020-69513-x Download
citation * Received: 30 March 2020 * Accepted: 09 July 2020 * Published: 28 July 2020 * DOI: https://doi.org/10.1038/s41598-020-69513-x SHARE THIS ARTICLE Anyone you share the following link
with will be able to read this content: Get shareable link Sorry, a shareable link is not currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt
content-sharing initiative